Filariasis (Wuchereria bancrofti and Brugia malayi)
Potential Severity
This chronic, debilitating infection can cause severe, disfiguring complications by obstructing lymphatic drainage.
Prevalence, Epidemiology, and Life Cycle
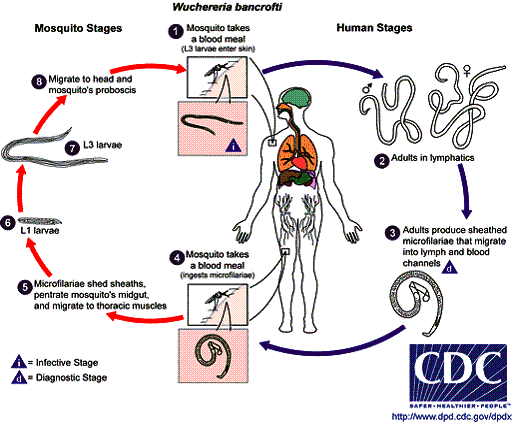
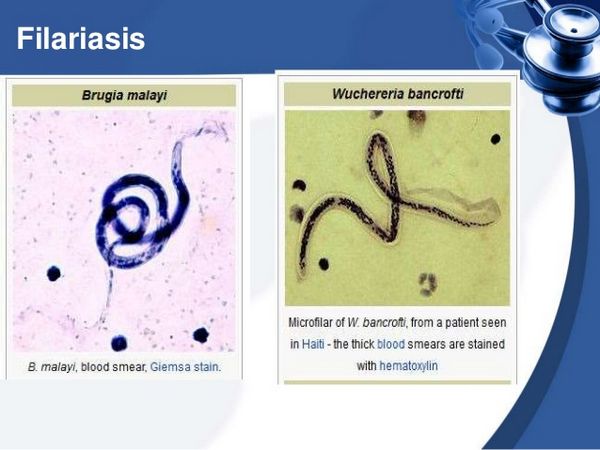
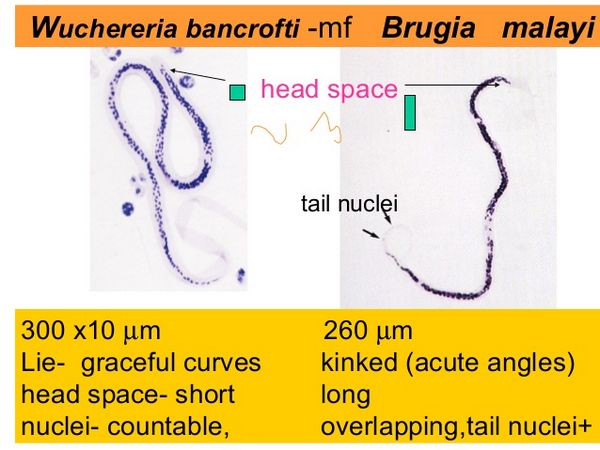
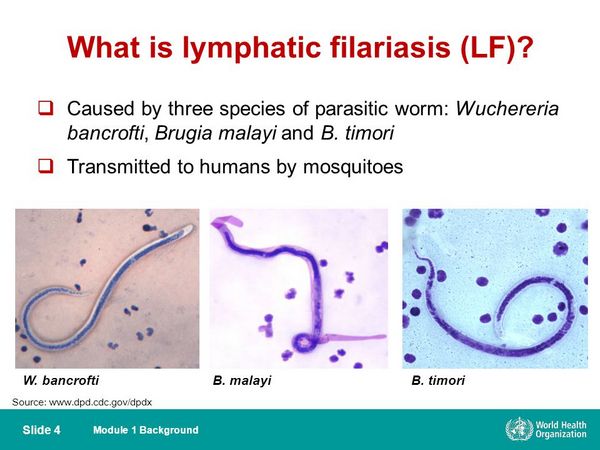
Microfilarial infection is less common than many other parasitic infections and is estimated to affect approximately 120 million people worldwide. Several worm strains can cause this disease. Wuchereria bancrofti is found throughout the tropics, and Brugia malayi is restricted to southern regions of Asia. A third strain, Brugia timori, is found only in Indonesia. Infectious larvae are transmitted through the bite of a mosquito.
Larvae pass from the skin into the lymphatic system, where they mature near the lymph nodes over several months. Adult worms, about 1.6 to 4 inches (40 to 100 mm) in length, can survive in the lymphatic system for 5 to 15 years. During this period, males and females mate and produce an average of 10,000 microfilariae daily (dimensions: 200 to 300 µm in length and 10 µm in width). The microfilariae are then released into the bloodstream.
The time from the initial insect bite to the appearance of microfilariae in the infected person is usually 12 months. In W. bancrofti, the highest concentration of microfilariae in the blood is generally found in the middle of the night, which explains why midnight blood smears are recommended for diagnosis. When a mosquito bites an infected person, it ingests microfilariae that develop into infective larvae over 10 to 14 days; these larvae can then be transmitted to a new human host. The percentage of mosquitoes containing infective larvae has been estimated to be just 1% in endemic areas.
Repeated mosquito bites are generally required to acquire this infection, which may help explain why adults-particularly men-more commonly develop symptomatic disease.
Clinical Presentation
Asymptomatic Filariasis
Many individuals have asymptomatic infection. Peripheral eosinophilia and palpable lymphadenopathy may be the only clinical manifestations. Children usually experience no symptoms, despite high numbers of microfilariae in their blood.
Inflammatory Filariasis
Adults more commonly develop strong allergic responses to worm invasion that begins approximately 1 year after exposure. Fever, chills, vomiting, headache, and malaise may be associated with lymphangitis of an extremity, orchitis, epididymitis, or scrotal swelling. The affected extremity becomes hot, swollen, erythematous, and painful, mimicking cellulitis. These symptoms are associated with peripheral leukocytosis and an increased percentage of eosinophils (6% to 25%). Unlike cellulitis, which usually begins peripherally and moves up the limb, inflammatory filariasis begins centrally near the lymph nodes and extends peripherally. Attacks may occur monthly and do not respond to antibiotics. The granulomatous response in the lymphatic tissue is thought to be a host inflammatory reaction to dying worms. Worm death is associated with the release of the rickettsial-like bacteria Wolbachia, which live in a symbiotic relationship within adult worms.
Obstructive Filariasis
Over time, chronic inflammation leads to fibrosis and permanent obstruction of lymphatic flow. This syndrome results from ongoing microfilarial infection. Persistent lymphatic obstruction and edema lead to marked skin thickening and deposition of collagenous material, eventually causing elephantiasis. Patients experience debilitating enlargement of the legs or massive enlargement of scrotal tissue, which makes walking difficult. Cellulitis caused by streptococci or Staphylococcus aureus may recur periodically and require antibiotic treatment. Rupture of lymphatics into the kidney or bladder can result in chyluria, and rupture into the peritoneum can cause chylous ascites.
| Clinical form | Key features | Typical findings |
|---|---|---|
| Asymptomatic filariasis | Infection without overt symptoms, often in children and young adults | Peripheral eosinophilia, palpable lymphadenopathy, high microfilarial burden on blood smear |
| Inflammatory filariasis | Acute inflammatory episodes beginning about 1 year after exposure | Fever, chills, malaise, painful lymphangitis, orchitis, epididymitis, scrotal swelling, leukocytosis with eosinophilia (about 6-25%) |
| Obstructive filariasis | Chronic lymphatic damage with progressive obstruction | Elephantiasis of limbs or scrotum, chronic edema, recurrent bacterial cellulitis, possible chyluria or chylous ascites |
Life Cycle of Wuchereria bancrofti and Brugia malayi
- Transmitted by the bite of an infected mosquito.
- Repeated mosquito bites are required.
- Microfilariae live in the lymphatic system, and worms enter the bloodstream at midnight (except in the South Pacific).
- Mosquitoes become infected by biting humans.
Diagnosis and Treatment
Giemsa- or Wright-stained peripheral smears should be obtained at midnight in all cases except those from the South Pacific. Identification of adult worms in the blood is definitive; however, worms are often not seen in early and late disease. Antibody and antigen assays are highly sensitive and specific. An IgG4 antibody titre correlates with active disease. An ELISA for W. bancrofti circulating antigen is now the diagnostic test of choice, and titres correlate with adult worm burden. A polymerase chain reaction (PCR) test for W. bancrofti has been developed, but it is not widely available. Biopsy of infected lymph nodes is generally not recommended, but when performed, it may reveal adult worms in addition to granuloma. Ultrasonography of dilated lymphatics in the spermatic cord has revealed motile worms. In early infection and during the inflammatory stage, peripheral eosinophilia is commonly seen.
| Test | Primary target | Typical use and notes |
|---|---|---|
| Midnight peripheral blood smear (Giemsa or Wright) | Circulating microfilariae | Definitive visualization of worms; best obtained at night for W. bancrofti; may be negative in early or late disease |
| Circulating antigen assays (ELISA) | W. bancrofti antigens | Highly sensitive and specific; titres correlate with adult worm burden and disease activity |
| Antibody assays (including IgG4) | Host antibody response | Supportive of infection; IgG4 levels tend to reflect active disease |
| PCR for filarial DNA | W. bancrofti genetic material | Very sensitive; limited availability; mainly used in reference or research settings |
| Ultrasound of dilated lymphatics | Adult worms in lymphatic channels | May demonstrate motile worms, especially in the spermatic cord or other dilated lymphatics |
| Lymph node biopsy | Adult worms and granulomatous reaction | Generally not recommended; may show worms and granuloma when performed for other indications |
| Peripheral eosinophil count | Eosinophilia | Common in early and inflammatory stages; nonspecific but supportive when combined with other findings |
Clinical Presentation of Filariasis
- Many people, particularly children, are asymptomatic.
- Inflammatory filariasis is associated with periodic erythema, warmth, pain, and swelling that mimic cellulitis (associated with peripheral eosinophilia).
- Obstructive disease results in chronic limb swelling (elephantiasis) because of lymphatic fibrosis.
- Obstructive disease can lead to recurrent bacterial cellulitis.
- Rupture of lymphatics can cause chyluria or chylous ascites.
- Release of the rickettsial-like bacteria Wolbachia from adult worms may be a major stimulus for inflammation.
Diagnosis and Treatment of Filariasis
- A midnight blood smear that demonstrates worms provides a definitive diagnosis.
- Worms may not be seen in early and late disease.
- Ultrasound of dilated lymphatics may demonstrate worms.
- Peripheral eosinophilia is common.
- Enzyme-linked immunosorbent assay is sensitive and specific, and levels correlate with disease activity.
- Diethylcarbamazine or ivermectin plus albendazole is used for treatment.
Treatment can exacerbate symptoms. During the chronic stages of disease, eosinophilia is generally not present. If worms cannot be identified, the diagnosis must be made on clinical grounds. A single dose of diethylcarbamazine is the recommended therapy, but it does not kill adult worms.
A reduction in the level of microfilariae in the blood is usually observed. Treatment may increase inflammation and may not halt progression to fibrosis and lymphatic obstruction. Ivermectin 200 to 400 mg/kg, combined with albendazole 400 mg, is another effective regimen that may more effectively kill adult worms.
In patients with more severe infection, a 3-week course of doxycycline kills the symbiont Wolbachia, resulting in sterility of adult worms. This treatment can be followed by diethylcarbamazine or ivermectin plus albendazole.
These agents typically exacerbate the host inflammatory reaction as microfilariae die, but eradication of Wolbachia with doxycycline eliminates this complication. Anti-inflammatory agents may be used to reduce the extent of inflammation, and elastic support stockings can help reduce moderate lymphedema. Long-term management is usually guided by clinicians experienced in tropical medicine and lymphatic disorders.