Trematodes
Essentials of Diagnosis
- Blood flukes: demonstration of eggs in feces, urine, or rectal biopsy
- Liver flukes: demonstration of eggs in feces
- Intestinal flukes: demonstration of adult worms or eggs in feces
- Lung flukes: demonstration of eggs in sputum or feces
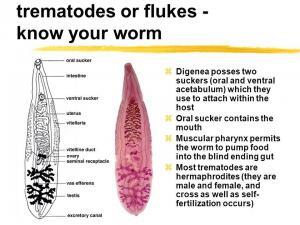
All trematode species that are parasitic in humans are digenetic. Sexual reproduction in the adult fluke is followed by asexual multiplication in the larval stage. Most adult trematode species have oral and ventral suckers. The life cycles of trematodes that are important human pathogens are similar among the five major species. Eggs are excreted in the feces of the host, hatch in fresh water, and require a primary host (snails) before infection occurs in humans. Trematode infections may be classified into four general groups, based on the final habitat of the adult flukes in humans: blood flukes, liver flukes, intestinal flukes, and lung flukes (Table 1).
General Considerations
Blood Fluke Infection (Schistosomiasis)
Epidemiology
Schistosomiasis is estimated to affect > 200 million people worldwide. It is endemic in Asia, Africa, South America, and other regions where the population is engaged in freshwater agriculture. The total number of infected individuals in the United States is estimated to exceed 500,000, and these cases are most often in immigrants from South America, Asia, and the Caribbean. The disease cannot spread in the United States because the specific snail that serves as an intermediate host does not exist in the United States. Unlike most other trematodes that are pathogenic in humans, adult schistosomes have separate sexes. The female fluke resides within a groove created by the fold of the lateral edges of the male fluke.
Each of the five blood fluke species that are pathogenic in humans has a specific geographic distribution: Schistosoma mansoni-Arabian Peninsula, Africa, the Caribbean, and South America; Schistosoma haematobium-the Middle East and Africa; Schistosoma japonicum-Japan, China, and the Philippines; Schistosoma mekongi-Southeast Asia; and Schistosoma intercalatum-West and Central Africa. The specific geographic distribution depends on the presence of the specific intermediate snail host.
Microbiology and Pathogenesis
The life cycle of the five major blood fluke species is as follows: adult flukes (1-2 cm in length) inhabit the venous system of the mesentery or urinary bladder; sexual reproduction results in characteristically shaped eggs that are excreted in the feces; the eggs hatch in fresh water, releasing ciliated, motile miracidia, which penetrate the body of the snail that serves as the intermediate host (a specific snail and geographic distribution exist for each blood fluke species); within the snail, the miracidia multiply asexually and, 4-6 weeks later, hundreds of fork-tailed cercariae emerge; the cercariae penetrate human skin, lose their tails, and become schistosomes that migrate to the lungs and liver, where maturation occurs; and ~ 6 weeks later, the mature adult fluke migrates to its final habitat in the venous system. The mean life span of adult flukes in humans is estimated to be 5-10 years.
Clinical Findings
The three stages of the blood fluke in humans (cercariae, mature flukes, and eggs) result in three major disease syndromes in chronological order: dermatitis (swimmers' itch), fever and constitutional symptoms (Katayama fever), and, finally, chronic fibro-obstructive disease (Table 2).
Signs and Symptoms
Dermatitis (swimmers' itch) is the acute form of schistosomiasis. It is characterized by a pruritic, papular rash that usually occurs within 1-3 days after cercarial penetration. Swimmers' itch rarely occurs after primary exposure and is more common in individuals who have been sensitized by earlier exposure.
Katayama fever occurs 4-8 weeks after skin penetration and coincides with egg production by the flukes. Katayama fever is most severe after S japonicum infection, but it occasionally occurs in patients infected with S mansoni and rarely in those infected with S haematobium. Patients experience an acute onset of fever, chills, headache, and cough. Physical findings often include lymphadenopathy and hepatosplenomegaly. Eosinophilia is common. Symptoms usually disappear within a few weeks after onset, but, rarely, death may occur, usually in association with S japonicum infection. Katayama fever may result from immune complex formation after a massive antigenic challenge from exposure to fluke eggs.
In chronic fibro-obstructive schistosomiasis, tissue damage occurs from a chronic inflammatory response to egg deposition. This process results in chronic granulomatous disease and eventual fibrosis. The liver and bowel are the sites most commonly affected in patients with infection caused by S mansoni, S japonicum, S mekongi, or S intercalatum. Patients often experience chronic abdominal pain and diarrhea. In the liver, fibrosis may result in portal hypertension, hepatosplenomegaly, esophageal varices, and variceal bleeding. Liver function tests usually remain normal for an extended period after infection. In chronic untreated cases, hepatic dysfunction, jaundice, ascites, and liver failure may occur.
In patients with S haematobium infection, the flukes are located in the vesicular plexus, and granuloma formation occurs in the bladder and ureters. Hematuria is the most common complaint. With prolonged infection, fibrosis, ureteral obstruction, and chronic renal failure may occur. The frequency of bladder cancer may be higher in patients with chronic S haematobium infection.
Pulmonary schistosomiasis may occur in patients with advanced liver cirrhosis and portal hypertension. The development of portosystemic shunts enables fluke eggs to bypass the liver and enter the pulmonary circulation. Patients with S haematobium may also develop pulmonary schistosomiasis. In these patients, eggs may enter the inferior vena cava from the vesicular plexus and then enter the pulmonary venous system.
Of patients infected with S japonicum, ~ 3% develop central nervous system schistosomiasis manifested by space-occupying lesions that may cause focal seizures. Granulomatous lesions that result from ectopic eggs in the vasculature of the spinal cord may rarely cause transverse myelitis.
Laboratory Findings
The most common laboratory findings in patients with schistosomiasis include eosinophilia, hematuria, anemia, and-in chronic end-stage infection-abnormal liver function tests, elevated serum creatinine, and uremia. The diagnosis is established by identifying characteristic schistosomal eggs in feces, urine, or a rectal biopsy specimen. Skin tests or serologic tests are helpful for epidemiologic studies but are not diagnostic of active infection.
Differential Diagnosis
Hepatic schistosomiasis may resemble any process that causes chronic hepatosplenomegaly and portal hypertension, including alcoholic cirrhosis, Wilson's disease, chronic hepatitis C infection, and many other conditions. S haematobium infection must be differentiated from bladder or ureteral cancer, reflux disease, and other syndromes that cause hematuria or chronic renal failure.
Treatment
Treatment for schistosomiasis is shown in Table 3. Praziquantel is safe and effective therapy for all five Schistosoma species that infect humans. Eggs may be shed for some time after treatment, and follow-up stool or urine examination should be obtained several months after therapy to ensure eradication.
Prognosis
The prognosis for schistosomiasis is excellent among patients who are treated before end-stage hepatic or renal disease develops. In these patients, prognosis depends on the severity of end-organ disease.
Prevention
The only effective means of prevention is to avoid contact with fresh water in areas where schistosomiasis is endemic.
Liver Fluke Infections (Clonorchiasis, Opisthorchiasis, and Fascioliasis)
General Considerations
The final habitat of liver flukes is the bile ducts. The liver flukes that most commonly cause human infection are Clonorchis sinensis (clonorchiasis), Opisthorchis spp. (opisthorchiasis), and Fasciola hepatica (fascioliasis).
C sinensis (Chinese or oriental liver fluke) is a flat, elongated fluke (~ 15 mm ( 3 mm) that inhabits the distal biliary capillaries. Humans are incidental hosts, and infection is endemic in China, Hong Kong, Korea, and Southeast Asia. Eggs eliminated in the feces are then ingested by a specific intermediate snail host. After ingestion, the eggs hatch into miracidia. The organisms multiply and produce extremely high numbers of cercariae that exit the snail and penetrate the skin of freshwater fish. Once the cercariae are on the fish skin, they encyst as metacercariae, which are infective in humans. Humans and fish-eating mammals acquire infection by ingesting raw or undercooked fish that contain metacercariae. After ingestion, metacercariae excyst in the human duodenum and pass through the ampulla of Vater, where the flukes mature into adults in the bile ducts.
O felineus and other species are endemic in Southeast Asia, Eastern Europe, and Russia. These parasites are common liver flukes of dogs and cats and may be transmitted to humans. The life cycle is similar to that of C sinensis.
F hepatica is a common liver fluke in sheep and cattle, and it is endemic in South America, Europe, Africa, Australia, and China. Humans are incidental hosts. Eggs are deposited in the biliary system and excreted in the feces. In fresh water, the eggs hatch, and the miracidia must reach and penetrate their specific snail host within 8 h. The cercariae emerge from snails and encyst on aquatic plants and sometimes in soil. The plants are consumed by humans, sheep, or cattle. The organisms excyst in the duodenum. The larvae penetrate through the intestinal wall into the peritoneum, enter through the liver capsule, and migrate to the bile ducts.
Clinical Findings
Most patients with C sinensis and O felineus infections are asymptomatic (Table 2). With heavy infection, biliary obstruction and cholangitis may occur. The frequency of cholangiocarcinoma may be increased in patients with chronic C sinensis infection. Diagnosis depends on demonstration of characteristic eggs in the feces.
Unlike clonorchiasis or opisthorchiasis, patients parasitized with F hepatica are usually symptomatic, and infection has two distinct clinical stages. In the early stage, symptoms correspond to hepatic migration of the larvae, and patients experience fever, right upper quadrant pain, hepatomegaly, and eosinophilia. Acute symptoms subside as the larvae enter the bile ducts, and most patients become asymptomatic in this later stage of infestation. Biliary obstruction, cholangitis, or biliary cirrhosis may occur rarely with heavy infestation. The definitive diagnosis is made by identifying characteristic eggs in the feces or bile. A positive serologic test suggests infection. Computed tomography of the liver may show small nodules and tortuous tracts created by the migrating larvae.
Differential Diagnosis
In the acute phase of F hepatica infection, patients may resemble those with acute cholangitis, cholecystitis, liver abscess, or Neisseria gonorrhoeae infection (Fitz-Hugh and Curtis syndrome). However, the presence of eosinophilia suggests F hepatica infection.
Treatment
Praziquantel is effective therapy for symptomatic patients with C sinensis or O felineus infection. Bithionol is the drug of choice for F hepatica infection (Table 3).
Prognosis
The prognosis for treated patients with liver fluke infection is excellent.
Prevention
Avoiding consumption of raw or undercooked freshwater fish is the only effective means of preventing C sinensis or O felineus infection. Individuals should avoid consuming undercooked aquatic plants and grasses in areas where F hepatica is endemic.
Intestinal Fluke Infection (Fasciolopsiasis)
General Considerations
The final habitat of intestinal flukes is the small bowel. Fasciolopsis buski is endemic in the Far East and Southeast Asia. Eggs are excreted in the feces, develop into cercariae in fresh water, and encyst on freshwater plants. After consumption by humans, the organisms excyst in the bowel and mature into adult flukes that measure 2-8 cm ( 1-2 mm. They reside in the upper portion of the small intestine, where they attach to the mucosa.
Clinical and Laboratory Findings
Most patients are asymptomatic, but heavy infection may cause abdominal pain, diarrhea, and occasionally malabsorption (Table 2). Diagnosis may be made by demonstrating characteristic eggs or adult flukes in the stool.
Differential Diagnosis
Most patients are asymptomatic. The differential diagnosis is that of patients with diarrhea and abdominal pain caused by bacteria and other parasites.
Treatment
Praziquantel is effective therapy for intestinal flukes (Table 3).
Prognosis
The prognosis is excellent.
Prevention
Individuals living in endemic areas should avoid consuming undercooked aquatic plants and grasses.

Lung Fluke Infection (Paragonimiasis)
General Considerations
Paragonimus westermani is endemic in the Far East, the Indian subcontinent, Central and South America, and West Africa. Human lung flukes produce eggs in sputum that are swallowed, excreted in the feces, and mature in fresh water into miracidia, which penetrate snails. Mature cercariae exit the snail, penetrate freshwater crayfish and crabs, and encyst. Humans acquire infection after ingesting raw, undercooked, or pickled freshwater crustacea. After ingestion, the organisms excyst in the duodenum, penetrate through the bowel wall, enter the peritoneal cavity, pass through the diaphragm into the pleural space, and enter the lungs, where they mature into flukes that measure 7-15 mm ( 5-8 mm. Ectopic eggs in the brain may result in space-occupying lesions and focal seizures.
Clinical Findings
Many patients with minimal to moderate infection are asymptomatic (Table 2). In acute infection, symptomatic patients experience cough productive of brown sputum or hemoptysis and eosinophilia. Heavy untreated infection may progress to chronic bronchitis or bronchiectasis with large-volume sputum production, pleuritic chest pain, pleural effusion, and lung abscess. Diagnosis is made by identifying characteristic eggs in the sputum or feces. Positive serology may be helpful in diagnosing ectopic infection. Diagnosis should also be suspected in individuals who do not live in endemic areas but have a history of consuming undercooked crustacea imported from endemic areas.
Differential Diagnosis
Pulmonary paragonimiasis resembles chronic bacterial infection, mycobacterial infection, or lung carcinoma. The presence of eosinophilia in patients who live in, or have travelled to, an endemic area should suggest paragonimiasis.
Treatment
Praziquantel is effective therapy (Table 3).
Prognosis
The prognosis is excellent in patients who are treated before chronic bronchitis or bronchiectasis develops.
Prevention
Individuals should avoid consuming raw or undercooked freshwater crustacea from areas where P westermani is endemic.
| Type | Infection | Species | Primary | Secondary | Parasitic Site in Humans |
|---|---|---|---|---|---|
|
Blood |
Schistosomiasis |
S mansoni |
Snails |
None |
Inferior mesenteric veins |
|
Liver |
Clonorchiasis |
C sinensis |
Snails |
Fish |
Bile ducts |
|
Intestinal |
Fasciolopsiasis |
F buski |
Snails |
Freshwater plants |
Small bowel |
|
Lung |
Paragonimiasis |
P westermani |
Snails |
Crabs and crayfish |
Lungs |
| Trematode | More Common | Less Common |
|---|---|---|
|
Blood flukes |
|
|
|
Liver flukes |
|
|
|
Intestinal flukes |
|
|
|
Lung flukes |
|
|
|
Species |
First Choice |
Second Choice |
|---|---|---|
|
S mansoni |
Praziquantel, 20 mg/kg twice daily during 1 day |
Oxamniquine, 15 mg/kg single dose. African-acquired infection, 20 mg/kg daily during 3 days |
|
S haematobium |
Praziquantel, 20 mg/kg twice daily during 1 day |
Metrifonate, 7.5 mg/kg single dose, weekly during 2 weeks |
|
S intercalatum |
Praziquantel, 20 mg/kg twice daily during 1 day |
No satisfactory alternative therapy |
|
S japonicum |
Praziquantel, 20 mg/kg three times daily during 1 day |
No satisfactory alternative therapy |
|
C sinensis |
Praziquantel, 25 mg/kg three times daily during 1 day |
No satisfactory alternative therapy |
|
F hepatica |
Bithionol 30-50 mg/kg on alternate days during 10-15 doses (maximum dose, 2 g/day) |
No satisfactory alternative therapy |
|
F buski |
Praziquantel, 25 mg/kg three times daily during 1 day Praziquantel, 25 mg/kg three times daily during 2 days |
No satisfactory alternative therapy |

















