Diflucan (Fluconazole)

Dosages
Diflucan 50 mg
| Quantity | Price per tablet | Total price | |
|---|---|---|---|
| 60 | C$1.31 | C$78.89 | |
| 90 | C$1.17 | C$105.19 | |
| 120 | C$1.10 | C$131.49 | |
| 180 | C$1.03 | C$185.47 | |
| 270 | C$0.99 | C$267.13 | |
| 360 | C$0.96 | C$344.64 |
Diflucan 100 mg
| Quantity | Price per tablet | Total price | |
|---|---|---|---|
| 30 | C$2.35 | C$70.59 | |
| 60 | C$1.78 | C$106.58 | |
| 90 | C$1.58 | C$142.56 | |
| 120 | C$1.49 | C$178.55 | |
| 180 | C$1.38 | C$249.14 | |
| 270 | C$1.32 | C$355.71 |
Diflucan 150 mg
| Quantity | Price per tablet | Total price | |
|---|---|---|---|
| 30 | C$3.00 | C$89.97 | |
| 60 | C$2.24 | C$134.26 | |
| 90 | C$1.98 | C$178.55 | |
| 120 | C$1.87 | C$224.22 | |
| 180 | C$1.74 | C$312.81 | |
| 270 | C$1.66 | C$447.06 |
Diflucan 200 mg
| Quantity | Price per tablet | Total price | |
|---|---|---|---|
| 30 | C$4.71 | C$141.18 | |
| 60 | C$3.41 | C$204.85 | |
| 90 | C$2.97 | C$267.13 | |
| 120 | C$2.76 | C$330.80 | |
| 180 | C$2.53 | C$455.37 | |
| 270 | C$2.39 | C$646.37 |
Diflucan 400 mg
| Quantity | Price per tablet | Total price | |
|---|---|---|---|
| 30 | C$7.84 | C$235.30 | |
| 60 | C$5.88 | C$352.95 | |
| 90 | C$5.24 | C$471.98 | |
| 120 | C$4.93 | C$591.01 | |
| 180 | C$4.60 | C$827.69 | |
| 270 | C$4.38 | C$1,182.02 |
Payment & Shipping
Your order is carefully packed and ships within 24 hours. Here is what a typical package looks like.
Sized like a regular personal letter (approximately 24x11x0.7 cm), with no indication of what is inside.



| Shipping Method | Estimated delivery |
|---|---|
| Express Free for orders over C$415.23 | Estimated delivery to Canada: 4-7 days |
| Standard Free for orders over C$276.82 | Estimated delivery to Canada: 14-21 days |








Discount Coupons
- Canada Day - July 1, 2026 10% CANADADAY10
- Boxing Day - December 26, 2026 12% BOXING12
Brand Names
| Country | Brand Names |
|---|---|
 Argentina Argentina | Candimicol Damicol Femixol Flucoginkan Fluconovag Fluzol Fungicil Fungocina Fungototal Honguil Plus Klonarizol Klonazol Micolis Novo Mutum Naxo C Nifurtox Niofen Periplum Ponaris Proseda F Triflucan |
 Australia Australia | Dizole Fluzole Ozole |
 Belgium Belgium | Candizole Fungimed |
 Brazil Brazil | Candix Candizol Celozol Farmazol Floltec Flucanil Flucanol Flucazol Flucocin Flucodan Flucoltrix Flucomed Fluconal Fluconax Fluconeo Fluconid Flucozen Flucozix Flunal Flunazol Fluotec Flusan Flutec Fungnon Glyflucan Helmicin Lertus Monipax Pantec Pronazol Riconazol Teczol Triazol Unizol Zelix Zolanix Zolmic Zolstatin Zoltec Zoltren |
 Czechia Czechia | Diflazon Fluco Forcan Mycomax Mycosyst Mykohexal |
 Denmark Denmark | Conasol Fungal |
 France France | Beagyne Triflucan |
 Germany Germany | Canex Canifug Fluco Fluc Flucobeta Flucoderm FlucoLich Flunazul Fungata |
 Greece Greece | Azoflu Azzol-S Dalrich Falipan Farviron Figalol Flucalit Flucocaps Flucodrug Flucofin Fluconapen Flucoran Flucozol Flukatril Flusenil Fluzomic Fumecal Funadel Fungo Fungram Fungustatin Fungusteril Fuxilidin Gynosant Hadlinol Medoflucon Mycazole Neomycol Opumyk Rifagen Stabilanol Tierlite Varmec Zidonil |
 Hungary Hungary | Dermyc Diflazon Flucohexal Flucoric Mycosyst Nofung |
 Italy Italy | Biozolene Elazor Flumicon Lefunzol Riflax Winch |
 Malaysia Malaysia | Avezol Biozole Flucon Fluconol Flucoric Flugal Fukole Medoflucon Odaft Stalene Zolstan |
 Mexico Mexico | Afungil Bioxel Candizol Difusel Fectrin Fharder Fleridux Flucoxan Fludisol Fluhexal Flukenol Flukezol Fluxes Fluxicap Fluzor Funser Lanfluzol Menifar Neofomiral Ongicil Oxifungol Ranflu Solarisol Terplex Waynazol Zilrin Zoldicam Zonal |
 Netherlands Netherlands | Flucoderm |
 New Zealand New Zealand | Canesten Fluconazole Flucazole |
 Poland Poland | Flucofast FlucoLEK Fluconazin Flumycon Mycomax Mycosyst |
 Portugal Portugal | Azoflune Fludocel Maxflin Reforce Supremase |
 Spain Spain | Citiges Lavisa Loitin Nesporac Solacap |
 Turkey Turkey | Biocanol Candidin Flucan Fluzole Fungan Kandizol Lumen Triflucan Trizol Zolax |
| Manufacturer | Brand Names |
|---|---|
| Cipla Limited | FCN Flucalup Forcan FZ |
| Inc. | FCN Flucalup Forcan FZ |
| Intas Pharmaceuticals Ltd. | FCN Flucalup Forcan FZ |
| Lupin Pharmaceuticals | FCN Flucalup Forcan FZ |
| Zenlabs Pharmaceutical Inc. | FCN Flucalup Forcan FZ |
FAQ
The health information contained herein is provided for educational purposes only. It is not intended to replace discussions with a healthcare professional. All decisions regarding patient care must be made by a healthcare professional, considering the unique characteristics of each patient.
The health information contained herein is provided for educational purposes only. It is not intended to replace discussions with a healthcare professional. All decisions regarding patient care must be made by a healthcare professional, considering the unique characteristics of each patient.
To find out more about Diflucan, please see our product information. No matter what type of treatment you use for a vaginal yeast infection, make sure to tell your doctor or other healthcare professional about any side effects you experience.
Diflucan is the only oral medication approved to treat yeast infections - it is 1 pill, 1 time. Diflucan has been proven to be as effective as 7 days of Monistat 7.
Description
Fluconazole is a widely used antifungal medication that helps treat yeast and other fungal infections by stopping fungus from growing in the body. It comes in different strengths and is often prescribed when an effective, convenient oral treatment is needed.
Uses
Fluconazole is used to treat oropharyngeal, esophageal, or vulvovaginal candidiasis, as well as other serious systemic Candida infections such as urinary tract infections, peritonitis, candidemia, disseminated candidiasis, meningitis, and pneumonia. It is also used to treat meningitis caused by Cryptococcus neoformans and to treat blastomycosis, coccidioidomycosis, and histoplasmosis.

Fluconazole has also been used for less severe infections, including superficial fungal infections, dermatophytoses, and onychomycosis. In addition, it is used to help prevent serious fungal infections such as coccidioidomycosis, cryptococcosis, and mucocutaneous candidiasis in people with human immunodeficiency virus (HIV) infection, and to prevent fungal infections in other people with weakened immune systems, such as cancer patients and bone marrow transplant patients.
Before starting fluconazole treatment, appropriate specimens for fungal culture and other relevant lab tests, such as serology and histopathology, should be collected to isolate and identify the organism causing the infection. Fluconazole treatment may be started while waiting for the results of these in vitro tests; however, once results are available, treatment should be adjusted as needed. If in vitro fluconazole susceptibility testing is done, the results should be interpreted cautiously, since currently available tests may not accurately reflect fluconazole's in vivo activity.
Candidal Infections
Oropharyngeal and Esophageal Candidiasis
Oral or IV fluconazole is used to treat oropharyngeal candidiasis in immunocompromised adults with acquired immunodeficiency syndrome (AIDS), advanced AIDS-related complex (ARC), cancer, or other severe underlying disease. It is also used orally or by IV to treat esophageal candidiasis in adults with AIDS, cancer, or other severe underlying disease, including progressive systemic sclerosis.
Fluconazole appears to be at least as effective as, and in some cases more effective than, other antifungal agents used for the initial treatment of oropharyngeal and/or esophageal candidal infections. It is considered a drug of choice for these infections.
Fluconazole has led to clinical resolution of the signs and symptoms of infection in 79-100% of patients with oropharyngeal candidiasis. However, microbiologic cures have generally been seen in 44-87% of patients, and relapse rates may be high, especially in neutropenic patients. In adults with esophageal candidiasis confirmed by endoscopy, fluconazole has produced clinical resolution of signs and symptoms in about 61-93% of patients. In one study of adults with esophageal candidiasis and progressive systemic sclerosis, fluconazole treatment produced mycologic cures in about 93% of patients within 2-4 weeks. Even so, the relapse rate was almost 100% within 3 months after fluconazole treatment was stopped.
HIV-infected patients with severe or recurring episodes of oropharyngeal or esophageal candidiasis may benefit from long-term suppressive or maintenance treatment (secondary prophylaxis) to help prevent relapse.
Some clinicians consider topical treatment with oral clotrimazole or oral nystatin to be the treatment of choice for uncomplicated oropharyngeal candidiasis in people with HIV, and recommend reserving systemic antifungal agents such as oral fluconazole, oral itraconazole, or oral ketoconazole for cases that do not respond to topical treatment and for severe oropharyngeal candidiasis with esophageal involvement. However, some clinicians prefer to use an oral azole antifungal as the initial treatment for oropharyngeal candidiasis.
Oral fluconazole or itraconazole oral solution is at least as effective as topical treatment. In a study of adults with HIV and oropharyngeal candidiasis, the response rate and mycologic eradication rate after 14 days of treatment were 100% and 75%, respectively, in those who received oral fluconazole (100 mg once daily), compared with 65% and 20% in those who received topical clotrimazole (10-mg oral lozenge 5 times daily). In another study, 14 days of treatment with oral fluconazole (100 mg once daily as an oral suspension) was more effective than 14 days of topical nystatin (500,000 units as an oral suspension 4 times daily). The mycologic cure rate was 60% in the fluconazole group and 6% in the nystatin group, and the relapse rate at day 42 was 27% and 11%, respectively. After long-term follow-up, the comparative efficacy of fluconazole and other antifungal agents still needs further study.
Systemic anti-infective treatment is necessary for esophageal candidiasis. Some clinicians state that a 14- to 21-day course of oral fluconazole (100 mg once daily) or itraconazole oral solution (200 mg once daily) is highly effective for treating esophageal candidiasis, and that oral ketoconazole is less effective. These clinicians suggest using an oral itraconazole solution in patients who do not respond to oral fluconazole. IV amphotericin B can be used in patients with otherwise refractory disease. In a randomized, multicentre, double-blind study comparing oral fluconazole (initial dosage 100 mg daily increased to 200 mg daily if no symptomatic improvement occurred within 1-2 weeks) for up to 8 weeks with oral ketoconazole (initial dosage 200 mg daily increased to 400 mg daily if no symptomatic improvement occurred within 1-2 weeks) in AIDS patients with endoscopically confirmed esophageal candidiasis, endoscopic cure and resolution of esophageal symptoms occurred in 91% and 85% of patients receiving fluconazole, respectively, while endoscopic cure and symptom resolution occurred in 52% and 65% of patients receiving ketoconazole, respectively. Results from another study in immunocompromised patients with esophageal candidiasis suggest that patients receiving fluconazole may need a shorter initial treatment course than those receiving ketoconazole.
Vulvovaginal Candidiasis
Oral fluconazole is used to treat uncomplicated vulvovaginal candidiasis and complicated vulvovaginal candidiasis in nonpregnant women. Before antifungal treatment is first used in a woman with signs and symptoms of uncomplicated vulvovaginal candidiasis, the diagnosis should be confirmed either by showing yeasts or pseudohyphae on direct microscopic examination of vaginal discharge (10% potassium hydroxide [KOH] wet mount or Gram stain) or by culture. Identifying Candida by culture in the absence of symptoms is not an indication for antifungal treatment, since about 10-20% of women carry Candida or other yeasts in the vagina. In women with recurrent vulvovaginal candidiasis, vaginal cultures should be obtained to confirm the diagnosis and identify unusual Candida species such as C. glabrata.
Uncomplicated Vulvovaginal Candidiasis
Oral fluconazole effectively treats uncomplicated vulvovaginal candidiasis when given as a single dose. A single 150-mg oral dose of fluconazole produces clinical cures, meaning the absence of vulvovaginal burning, itching, swelling, erythema, excoriation, dyspareunia, and/or ulceration, together with a substantial decrease in vaginal discharge, 5-16 days after the dose in about 90-100% of nonpregnant women with uncomplicated vulvovaginal candidiasis. Mycologic cures occur in about 77-100%. At 27-62 days after the single dose, clinical and mycologic cure rates are 61-90%, and the rate of relapse, reinfection, or recolonization is about 23%. Results from several studies in patients with uncomplicated vulvovaginal candidiasis suggest that a single 150-mg oral dose of fluconazole is as effective as multiple-dose regimens of intravaginal clotrimazole, econazole, miconazole, or terconazole. In addition, a single oral dose of fluconazole is as effective as oral itraconazole or oral ketoconazole for this condition.
In controlled studies of patients with vulvovaginal candidiasis, clinical and mycologic cure rates at 14 days and at 30-35 days were similar in patients receiving oral fluconazole (given as a single 150-mg dose) and those receiving intravaginal clotrimazole (given as a 100-mg vaginal tablet once daily for 7 days) or miconazole (given as a 100-mg vaginal cream once daily for 7 days). At 14 days, the clinical cure rate was reported to be about 95-96% with fluconazole and 95-97% with intravaginal clotrimazole or miconazole, and the mycologic cure rate was reported to be 77-80% with fluconazole and 72-82% with intravaginal clotrimazole or miconazole. At 30-35 days, the clinical cure rate was reported to be about 69-75% with fluconazole and 72-80% with intravaginal clotrimazole or miconazole, and the mycologic cure rate was reported to be 61-63% with fluconazole and 57-63% with intravaginal clotrimazole or miconazole.
Canadian clinical guidance generally recommends treating uncomplicated vulvovaginal candidiasis, defined as infection that is mild to moderate, sporadic or infrequent, most likely caused by C. albicans, or occurring in immunocompetent women, with an intravaginal azole antifungal such as butoconazole, clotrimazole, miconazole, terconazole, or tioconazole given as an appropriate single-dose or short-course regimen, or with a single oral dose of fluconazole. These regimens have generally been associated with clinical and mycologic cure rates of 80-90% in otherwise healthy, nonpregnant women with uncomplicated infections. Some clinicians suggest that a single oral dose of fluconazole may offer an advantage over conventional intravaginal treatment because it can help support adherence and may reduce or eliminate concurrent rectal infections that may serve as a source of reinfection. When weighing the potential risks and benefits of oral versus intravaginal treatment, the possibility of toxicity such as hepatotoxicity and drug interactions with oral treatment should be considered. Side effects are reported more often in patients receiving a single oral dose of fluconazole than in those receiving intravaginal treatment, and that should also be weighed carefully.
Complicated and Recurrent Vulvovaginal Candidiasis
Oral fluconazole is used to treat complicated vulvovaginal candidiasis, including recurrent and severe infections. Complicated vulvovaginal candidiasis is defined as infections that are recurrent or severe, caused by Candida other than C. albicans, or occurring in pregnant women or women with underlying conditions such as uncontrolled diabetes, debilitation, or immunosuppression.
Optimal regimens for recurrent vulvovaginal candidiasis, usually defined as 4 or more episodes of symptomatic vulvovaginal candidiasis each year, have not been established. Although each episode caused by C. albicans may respond to the usual single-dose oral fluconazole regimen or short-course intravaginal antifungal treatment, a longer initial treatment course may be needed to achieve mycologic remission, and long-term maintenance treatment may be needed to prevent relapse. Canadian clinicians generally use an initial intensive regimen of 7-14 days of an intravaginal azole antifungal or a 2-dose regimen of oral fluconazole (150 mg repeated 3 days later), followed by a maintenance antifungal regimen for 6 months. For maintenance, commonly used options include intravaginal clotrimazole (500 mg once weekly), oral ketoconazole (100 mg once daily), oral fluconazole (100-150 mg once weekly), or oral itraconazole (400 mg once monthly or 100 mg once daily). These maintenance regimens can effectively reduce recurrent infections; however, 30-40% of women will have recurrent disease once maintenance treatment is stopped.
The response rate to short-course antifungal regimens is lower in patients with severe vulvovaginal candidiasis, meaning extensive vulvar erythema, edema, excoriation, and fissure formation. Either a 2-dose regimen of oral fluconazole (150 mg repeated 3 days later) or 7-14 days of an intravaginal azole antifungal is recommended for these infections. These longer regimens may also be needed to treat vulvovaginal candidiasis in women with underlying debilitating medical conditions, such as uncontrolled diabetes or corticosteroid therapy. Vulvovaginal candidiasis may occur more often and may be more severe in women with human immunodeficiency virus (HIV) infection than in women without HIV. These infections have been recognized as an early manifestation of acquired immunodeficiency syndrome (AIDS) in women. While optimal treatment for recurrent vulvovaginal candidiasis in women with HIV has not been established, there is currently no evidence that these women have a lower response rate to the intravaginal or oral antifungal regimens usually recommended for vulvovaginal candidiasis. Therefore, Canadian guidance generally recommends treating vulvovaginal candidiasis in women with HIV in the same way as in women without HIV infection.
Recurrent vulvovaginal candidiasis may rarely be caused by resistant strains of C. albicans or, more commonly, by other Candida species with reduced susceptibility to azole antifungals such as C. glabrata. It has been suggested that repeated treatment of recurrent vulvovaginal candidiasis with intravaginal azole antifungals, along with widespread and/or inappropriate self-treatment, may favour the selection of Candida strains that are resistant to azole antifungals. The best treatment for vulvovaginal candidiasis caused by Candida with reduced susceptibility to azole antifungals has not been clearly established. Canadian guidance generally recommends 7-14 days of therapy with a non-fluconazole antifungal for vulvovaginal candidiasis caused by non-albicans Candida species. In some cases, intravaginal boric acid, typically 300-600 mg once daily for 14 days, may be considered, and referral to a specialist is appropriate if symptoms recur or treatment is unsuccessful.
Candidemia and Other Candidal Infections
Fluconazole has effectively treated serious candidal urinary tract infections, peritonitis, and pneumonia. It has also been effective in treating chronic mucocutaneous candidiasis, candidemia, chronic disseminated candidiasis (hepatosplenic candidiasis), candidal endocarditis, candidal meningitis, candidal osteomyelitis, and other severe systemic candidal infections. It has also been effective in treating some candidal infections that did not respond to amphotericin B. Fluconazole has been effective in treating life-threatening candidal infections in organ transplant patients receiving immunosuppressive treatment. In some renal allograft recipients, fluconazole treatment effectively cleared fungal infections without stopping or reducing the dosage of immunosuppressive treatment.
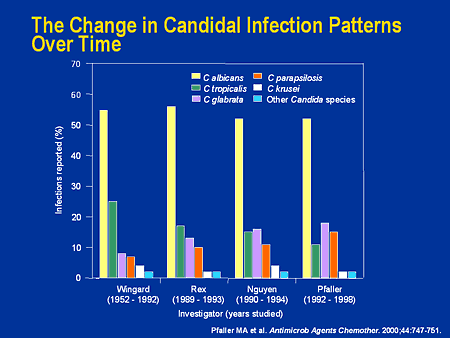
Both IV amphotericin B and IV or oral fluconazole are considered drugs of choice for invasive systemic candidiasis. However, the best antifungal regimens for these infections have been difficult to identify, and each drug has drawbacks. While fluconazole may be better tolerated and easier to give than IV amphotericin B, fluconazole-resistant strains of C. albicans are being isolated with increasing frequency in patients who have previously received fluconazole, especially those with HIV. Some candidal infections, such as candidemia, are increasingly caused by strains that are intrinsically resistant to fluconazole, such as C. krusei, or are likely to be fluconazole-resistant, such as C. glabrata. The choice of antifungal for the initial treatment of invasive candidal infections should take into account local and/or institutional epidemiologic data on the prevalence of different candidal strains and their resistance patterns, the patient's colonization status, the severity and duration of neutropenia or immunosuppression, and any previous fluconazole use. Most clinicians recommend IV amphotericin B if the infecting organism is known or likely to be C. krusei; however, fluconazole is preferred if the infection is caused by C. lusitaniae.
Because candidemia is associated with substantial morbidity and a risk of long-term complications, antifungal treatment is generally recommended for all patients with candidemia, whether neutropenic or nonneutropenic, in addition to removal and/or replacement of any intravascular catheters. It has been suggested that oral fluconazole may be preferred over IV amphotericin B for treating candidemia in nonneutropenic patients, whether stable or unstable, unless there is evidence that the infection is caused by fluconazole-resistant strains or the patient has previously received fluconazole. IV amphotericin B is generally preferred for severe candidemia in patients whose infections may be caused by fluconazole-resistant strains such as C. krusei or C. glabrata, in patients who have recently received fluconazole, and in immunocompromised patients such as those with HIV infection. In controlled studies in patients with candidemia, responses to conventional IV amphotericin B therapy and fluconazole therapy were similar, and both regimens appeared to be effective. A prospective, randomized study that included non-neutropenic and neutropenic patients with documented or presumed invasive candidiasis also found similar overall response rates with conventional amphotericin B and fluconazole. However, additional study is needed to more fully evaluate the relative efficacy of these agents in immunocompromised patients or in those with severe infections, and in Canadian practice treatment selection should be based on the clinical setting and specialist judgement.
Oral fluconazole or IV amphotericin B is recommended when treatment of candiduria is indicated, such as in symptomatic or neutropenic patients, low-birthweight infants, renal allograft recipients, or patients who will undergo urologic procedures. Fluconazole may be preferred unless the infection is caused by C. krusei or C. glabrata. Bladder irrigation with conventional amphotericin B has also been used. In a randomized study in hospitalized geriatric patients with funguria caused by C. albicans, C. tropicalis, or C. glabrata, 5 days of bladder irrigation with conventional amphotericin B (25 mg of amphotericin B in 500 mL of 5% dextrose injection infused through an indwelling bladder catheter at a rate of 42 mL/hour) or a 5-day course of oral fluconazole (200-mg loading dose on day 1 followed by 100 mg once daily for four additional days) eradicated the funguria in 83% and 66% of patients, respectively.
Fluconazole is also used prophylactically to reduce the incidence of candidiasis in bone marrow transplant recipients who are receiving chemotherapy or radiation therapy. It has also been used for primary prophylaxis against fungal infections, including candidiasis, in a limited number of patients considered at high risk of developing these infections, such as neutropenic cancer patients, patients colonized with Candida and/or receiving corticosteroids, and certain patients with AIDS.
Fluconazole has been used with good results in several patients with endophthalmitis caused by Candida. However, treatment failures have been reported, and the role of the drug in treating this infection remains unclear. Studies in rabbits indicate that fluconazole is distributed into the eye and that it inhibits the growth of C. albicans in rabbit choroid-retina tissue and vitreous body when IV treatment is started within 24 hours after inoculation. The drug did not effectively inhibit growth of the organism when IV treatment was started 7 days after inoculation, when the infection was already well established.
Cryptococcal Infections
Oral or IV fluconazole is used in immunocompetent or immunocompromised adults to treat meningitis caused by C. neoformans. It has been effective for the initial treatment of acute cryptococcal meningitis in both adults with HIV and adults without HIV. It has produced clinical resolution of signs and symptoms of infection in about 34-75% of these patients. Although amphotericin B, with or without concomitant flucytosine, has been considered the initial treatment of choice for cryptococcal meningitis, fluconazole is an alternative in patients whose disease is not severe because it is generally well tolerated and reaches high concentrations in CSF. While experience in children is limited, fluconazole may also be considered as an alternative to amphotericin B in this age group.
Fluconazole has been effective in treating acute cryptococcal meningitis in some patients who did not respond to amphotericin B. However, some evidence suggests that fluconazole may be less effective than amphotericin B in the early treatment of acute cryptococcal meningitis in patients with AIDS and may clear the CSF more slowly. In a randomized, multicentre study comparing amphotericin B (mean dosage of 0.4-0.5 mg/kg/day for 10 weeks with or without flucytosine) with oral fluconazole (400 mg on the first day and 200 to 400 mg/day after that for 10 weeks) in patients with AIDS and cryptococcal meningitis, treatment was effective in 40% of patients receiving amphotericin B and 34% of those receiving fluconazole. Although overall mortality was similar in the two groups (14% with amphotericin B versus 18% with fluconazole), mortality during the first 2 weeks of treatment was higher in patients receiving fluconazole (15% versus 8% with amphotericin B). CSF cultures remained positive for an average of about 42 days in patients receiving amphotericin B and 64 days in those receiving fluconazole. In another study comparing amphotericin B (0.7 mg/kg/day for 1 week, followed by the same dose 3 times weekly for 9 weeks combined with flucytosine 150 mg/kg/day) with oral fluconazole (400 mg/day for 10 weeks) in a small number of patients with AIDS and cryptococcal meningitis, initial treatment was effective in all patients receiving amphotericin B but in only 43% of those receiving fluconazole; CSF cultures remained positive for an average of about 16 and 41 days, respectively. Although the patient groups in this study were similar in terms of how severe the cryptococcal infection was, the helper/inducer (CD4+, T4+) T-cell count was lower in the fluconazole group, which makes the results harder to interpret.
Many clinicians recommend starting treatment of cryptococcal meningitis in HIV-infected patients with IV amphotericin B (with flucytosine) for at least 2 weeks, or until the patient's condition has stabilized, followed by oral fluconazole or oral itraconazole for at least another 8-10 weeks or longer. Until more data and clinical experience are available, some clinicians believe that starting with IV amphotericin B (with or without flucytosine) followed by fluconazole maintenance therapy is the most cautious approach, especially in patients with more severe disease and those at higher risk. They also suggest that fluconazole as initial therapy should probably be reserved for patients who do not respond adequately to amphotericin B, cannot tolerate it, or have less severe disease (for example, no neurologic symptoms and low CSF cryptococcal antigen titres).
The relative efficacy of initial treatment with conventional IV amphotericin B given with flucytosine (100 mg/kg/day) or placebo for 2 weeks, followed by oral fluconazole (800 mg/day for 2 days, then 400 mg/day for 8 weeks) or oral itraconazole (600 mg/day for 3 days, then 400 mg/day for 8 weeks), has been studied in a double-blind multicentre trial in patients with AIDS-associated cryptococcal meningitis. At 2 weeks, CSF cultures were negative in 60% of those who received amphotericin B with flucytosine compared with 51% of those who received amphotericin B alone. The clinical response to oral fluconazole or oral itraconazole as follow-up treatment was similar. Still, the rate of CSF sterilization at 10 weeks was higher in those who received fluconazole (72%) than in those who received itraconazole (60%).
A regimen of oral fluconazole (400 mg/day for pulmonary infections or 400-800 mg/day for CNS infections) and oral flucytosine (100-150 mg/kg/day) has been used as an alternative treatment for pulmonary or CNS cryptococcal infections in a limited number of patients. Although fluconazole with flucytosine may be effective for mild to moderate pulmonary cryptococcal infections, this regimen has been ineffective in some patients with cryptococcal meningitis. It is not recommended as initial treatment for these infections. In addition, when used to treat cryptococcal meningitis in HIV-infected individuals, a regimen of fluconazole plus flucytosine has been linked to a high rate of side effects, leading to flucytosine being stopped in 28% of patients.
The mortality rate in patients with AIDS during the first episode of cryptococcal meningitis has been about 25-58% despite amphotericin B treatment. In patients with AIDS who respond to initial antifungal therapy, the relapse rate for cryptococcal infection is 35-65%. Chronic maintenance or suppressive antifungal therapy is generally considered necessary after initial treatment unless immune recovery has occurred with potent antiretroviral therapy.
Oral fluconazole has been effective when used as long-term maintenance therapy to prevent relapse of cryptococcal meningitis in patients with AIDS. Results of a multicentre study comparing the safety and efficacy of oral fluconazole (200 mg once daily) with IV amphotericin B (1 mg/kg once weekly) for preventing relapse in patients with AIDS who had negative cryptococcal cultures after adequate initial amphotericin B treatment indicate that the fluconazole regimen was more effective at preventing relapse of culture-positive meningitis and was better tolerated than the amphotericin B regimen for maintenance therapy in these patients. In a multicentre study comparing oral fluconazole maintenance (100-200 mg/day) with placebo in these patients, the overall calculated cumulative risk of cryptococcal recurrence at any site after 1 year of fluconazole maintenance was 5% versus 100% with placebo.
The effectiveness of fluconazole maintenance in preventing cryptococcal relapse in patients with persistent prostatic infection after primary antifungal therapy appears to be lower than in patients overall. Still, this reduced effectiveness may be partly overcome by increasing the dosage. In a limited number of adults with AIDS who had persistent urinary cryptococcosis, including prostate infection, after adequate primary amphotericin B therapy (with or without flucytosine) for cryptococcal meningitis, oral fluconazole maintenance therapy resulted in mycologic cure in most patients. In contrast, relapse of cryptococcosis (with prostatic massage) and/or systemic infection occurred in the remaining patients during fluconazole maintenance.
Response in these patients, defined as sustained suppression of Cryptococcus after prostatic massage and no evidence of systemic or CNS relapse, appears to be dose-dependent and generally requires oral fluconazole 200 to 600 mg/day. It may take several weeks to months of maintenance therapy before a response becomes apparent; in one study, the probability of response was estimated to be 36% after 4 weeks and 59% after 27 weeks of maintenance. Relatively high dosages, such as 600 mg/day or more, may effectively sterilize multiple large prostatic abscesses in some patients. However, factors that may predict response and the best antifungal therapy for these patients still need to be clarified. Because of the risk of relapse, some clinicians also caution that a prostatic focus of infection should be adequately ruled out in any male patient before low-dose fluconazole maintenance is considered.
Oral fluconazole has been effective in a limited number of patients for treating cutaneous or subcutaneous cryptococcosis. The drug has also been effective for treating cryptococcal pneumonia in a limited number of patients, although some patients required surgical removal of the focus of infection. However, cryptococcal pneumonia may be a sign of disseminated infection and that possibility should be considered.
Coccidioidomycosis
Oral fluconazole has been used successfully to treat coccidioidomycosis caused by Coccidioides immitis, including meningitis, pulmonary infections, and disseminated infections involving soft tissue or bone and joint. In adults with coccidioidal meningitis, fluconazole has produced clinical and/or laboratory signs of improvement when used alone or together with amphotericin B. Oral fluconazole has been used to treat coccidioidal meningitis in both HIV-infected and HIV-negative individuals. Because fluconazole is generally well tolerated and has favourable pharmacokinetics, such as reaching high concentrations in CSF after oral or IV use, it is considered a less toxic alternative to amphotericin B for coccidioidal meningitis and other persistent coccidioidal infections, especially because these infections usually require long-term antifungal therapy. Fluconazole is considered a drug of choice for coccidioidomycosis; however, IV amphotericin B is generally preferred for the initial treatment of severe coccidioidomycosis, especially in immunocompromised patients, including those with HIV. More study is needed to assess efficacy, establish the best oral fluconazole dosage for coccidioidomycosis, and determine whether the drug substantially reduces illness and death. Some data suggest that response rates may improve with higher fluconazole dosages, that is, 400 mg/day or more. It has been suggested that relatively low dosages, such as 50-100 mg/day, may increase the risk of recurrence.
Blastomycosis
Oral fluconazole has been used to treat blastomycosis caused by Blastomyces dermatitidis in North America, including Canada. While oral itraconazole or IV amphotericin B are considered drugs of choice for blastomycosis, oral fluconazole or oral ketoconazole are considered alternatives. IV amphotericin B is generally preferred for severe infections, especially those involving the CNS, and for the initial treatment of presumed blastomycosis in immunocompromised patients, including people living with HIV.
There is some evidence that oral itraconazole may be more effective than oral fluconazole or oral ketoconazole for treating blastomycosis. Many clinicians consider itraconazole the preferred azole antifungal for nonmeningeal, non-life-threatening blastomycosis and also recommend it as follow-up therapy in patients with more severe infections after an initial response to IV amphotericin B. When choosing an antifungal for patients with blastomycosis, it is important to consider that treatment failures have been reported when an oral antifungal, such as ketoconazole, was used in patients with cutaneous or pulmonary blastomycosis who also had asymptomatic or subclinical CNS involvement at the time of diagnosis.
Histoplasmosis
Oral fluconazole has been used with some success to treat histoplasmosis caused by Histoplasma capsulatum. While IV amphotericin B or oral itraconazole are considered drugs of choice for histoplasmosis, oral fluconazole or oral ketoconazole are considered alternatives. Oral fluconazole (400-800 mg/day) has been effective in a limited number of HIV-infected patients with mild or moderately severe disseminated histoplasmosis; however, IV amphotericin B is generally preferred for the initial treatment of severe, life-threatening histoplasmosis, especially in immunocompromised patients such as those with HIV. In addition, oral itraconazole is generally the preferred azole antifungal for mild to moderate histoplasmosis or as follow-up therapy after a response has been achieved with amphotericin B in severe infections.
Sporotrichosis
Fluconazole is used as an alternative treatment for sporotrichosis. IV amphotericin B is usually considered the treatment of choice for the initial management of severe, life-threatening infections and whenever there is CNS involvement. Oral itraconazole is considered the treatment of choice for cutaneous, lymphocutaneous, or mild pulmonary or osteoarticular sporotrichosis, and for follow-up therapy in more severe infections after a response has been achieved with IV amphotericin B. Fluconazole is considered second-line treatment for cutaneous, lymphocutaneous, or osteoarticular sporotrichosis and, because it may be less effective than itraconazole, should be used only if the patient cannot tolerate itraconazole. Some clinicians state that fluconazole is not effective and should not be used for pulmonary sporotrichosis.
Aspergillosis
Fluconazole has been used orally, by IV, or by intracavitary infusion in a few adults to treat pneumonia or other respiratory tract infections caused by Aspergillus fumigatus, A. niger, or A. terreus. In one study, fluconazole had a clinical efficacy rate of 23-53% in Aspergillus infections and a mycologic cure rate of about 50% for A. fumigatus infections. Fluconazole has shown mixed results in treating Aspergillus infections. IV amphotericin B is generally considered the treatment of choice, and itraconazole is usually the preferred alternative for aspergillosis.
Dermatophytoses
Oral fluconazole has been effective in treating certain dermatophytoses, such as tinea capitis, tinea corporis, tinea cruris, and tinea pedis, caused by Epidermophyton, Microsporum, or Trichophyton. Oral fluconazole has also been effective for treating pityriasis (tinea) versicolor and onychomycosis.
Oral fluconazole (3-6 mg/kg/day for 2-6 weeks) has effectively treated tinea capitis in children 1.5-16 years of age and has resulted in clinical and mycologic cure in about 88-90% of patients. For tinea corporis, tinea cruris, or tinea pedis in adults, oral fluconazole has been effective in a once-weekly regimen (150 mg once weekly for 2-6 weeks). There is evidence that this once-weekly regimen is as effective as a once-daily regimen of the drug (50 mg once daily) for these infections. Results of a randomized study indicate that the eradication rate at the end of treatment in patients with tinea corporis or tinea cruris is 82-88% in those receiving the once-weekly regimen and 94-100% in those receiving the once-daily regimen; at 1-month follow-up, the overall eradication rates were 91-100% and 91-94%, respectively. Although the best oral fluconazole dosage regimen for onychomycosis has not been established, a once-weekly regimen (150-450 mg once weekly for 3-12 months) has been effective in a limited number of adults with toenail onychomycosis. However, more study is needed, and some evidence suggests that fluconazole may be less effective than oral itraconazole or oral terbinafine for onychomycosis.
Tinea corporis and tinea cruris can usually be treated effectively with a topical antifungal. However, an oral antifungal may be needed if the disease is extensive, dermatophyte folliculitis is present, the infection is chronic or does not respond to topical treatment, or the patient is immunocompromised or has another coexisting condition. Tinea capitis and tinea barbae are generally treated with an oral antifungal. While topical antifungals are usually enough for uncomplicated tinea manuum and tinea pedis, oral treatment is usually needed for hyperkeratotic areas of the palms and soles, chronic moccasin-type (dry-type) tinea pedis, and tinea unguium (onychomycosis).
Prevention of Fungal Infections in HIV-infected Individuals
Fluconazole has been used in HIV-infected individuals for primary prophylaxis against serious fungal infections, such as cryptococcosis, and for long-term suppressive or maintenance therapy (secondary prophylaxis) to prevent recurrence or relapse of certain fungal infections, such as coccidioidomycosis, cryptococcosis, and mucocutaneous candidiasis.
In Canada, guidance on preventing opportunistic infections in people living with HIV includes recommendations on reducing exposure to opportunistic pathogens, preventing first episodes of disease, and preventing recurrence. Canadian guidance generally does not recommend primary prophylaxis to prevent first episodes of mucocutaneous candidiasis in HIV-infected adults, adolescents, infants, and children. While routine primary prophylaxis to prevent first episodes of coccidioidomycosis, cryptococcosis, or histoplasmosis in HIV-infected adults, adolescents, infants, and children is not generally recommended, primary prophylaxis against cryptococcosis or histoplasmosis may be considered in selected individuals. Canadian practice also supports long-term suppressive or maintenance therapy (secondary prophylaxis) for HIV-infected adults, adolescents, infants, and children who have completed initial treatment for documented coccidioidomycosis, cryptococcosis, or histoplasmosis to help prevent recurrence or relapse. In addition, HIV-infected individuals who have frequent or severe recurrences of mucocutaneous candidiasis (oropharyngeal, esophageal, vaginal) may benefit from long-term suppressive or maintenance therapy.
Because of concerns about the use of oral azole antifungals during pregnancy, fluconazole should not be used for primary prophylaxis or for chronic suppressive or maintenance therapy in pregnant women. If a woman becomes pregnant while receiving fluconazole prophylaxis and chooses to continue the pregnancy, prophylaxis should be stopped. Effective contraception is recommended for all HIV-infected women receiving an oral azole antifungal for suppressive therapy. Conventional IV amphotericin B may be the preferred option if long-term suppressive or maintenance therapy against coccidioidomycosis, cryptococcosis, or histoplasmosis is needed in an HIV-infected pregnant woman, especially during the first trimester.
Primary Prophylaxis
Coccidioidomycosis
The United States Public Health Service/Infectious Diseases Society of America (USPHS/IDSA) states that routine primary prophylaxis against coccidioidomycosis in HIV-infected patients living in endemic areas is not recommended. Some clinicians suggest that primary prophylaxis should be considered for HIV-infected individuals who have positive CF serologic results without active disease. Still, it is unclear whether primary prophylaxis would be helpful for HIV-infected individuals living in areas where coccidioidomycosis is endemic. Active coccidioidomycosis has developed in HIV-infected individuals who were receiving azole therapy for other conditions. Routine skin testing with coccidiosis in HIV-infected individuals who live in areas endemic for coccidioidomycosis is not predictive of disease and generally is not recommended. Within an endemic area, a positive serologic test might indicate an increased risk of active infection; however, routine serologic testing does not appear to be helpful or recommended.
Cryptococcosis
The United States Public Health Service/Infectious Diseases Society of America (USPHS/IDSA) states that, although routine primary prophylaxis against cryptococcosis is not recommended, it may be considered in HIV-infected adults and adolescents with CD4+ T-cell counts below 50/mm3 and in infants and children with severe immunosuppression, as defined by age-adjusted criteria. Routine prophylaxis is not recommended because cryptococcal disease is relatively uncommon, there is no evidence that prophylaxis improves survival, and there are concerns about drug interactions, the potential development of resistance, and cost. The need for primary prophylaxis or suppressive therapy against other fungal infections, such as coccidioidomycosis, histoplasmosis, or mucocutaneous candidiasis, should be taken into account when deciding about primary prophylaxis against cryptococcosis. Routine serum cryptococcal antigen testing in people without symptoms is not recommended because the results are unlikely to affect treatment decisions. HIV-infected individuals cannot completely avoid exposure to Cryptococcus neoformans; there is no evidence that exposure to pigeon droppings increases the risk of cryptococcosis.
Oral fluconazole is the treatment of choice for primary prophylaxis against cryptococcosis in HIV-infected adults, adolescents, infants, and children, and itraconazole given as oral capsules is considered an alternative.
Histoplasmosis
The United States Public Health Service/Infectious Diseases Society of America (USPHS/IDSA) states that primary prophylaxis against histoplasmosis may be considered in HIV-infected adults or adolescents with CD4+ T-cell counts below 100/mm3 who are at especially high risk of exposure to Histoplasma capsulatum because of occupational exposure or because they live in a community with a hyperendemic histoplasmosis rate of at least 10 cases/100 patient-years. It may also be considered for HIV-infected infants or children with severe immunosuppression who live in areas where histoplasmosis is endemic. When deciding whether to use primary prophylaxis against histoplasmosis in these HIV-infected individuals, clinicians should consider the local incidence of histoplasmosis, the possibility of drug interactions, toxicity, development of resistance, cost, and the need for prophylaxis against other fungal infections such as candidiasis or cryptococcosis.
The treatment of first choice for primary prophylaxis against histoplasmosis in HIV-infected adults, adolescents, or pediatric patients is oral itraconazole given as capsules; the USPHS/IDSA makes no recommendation regarding an alternative to itraconazole. There is some evidence that oral fluconazole may be ineffective in preventing first episodes of histoplasmosis.
Mucocutaneous Candidiasis
The United States Public Health Service/Infectious Diseases Society of America (USPHS/IDSA) states that primary prophylaxis to prevent first episodes of mucocutaneous candidiasis (esophageal, oropharyngeal, vaginal) in HIV-infected adults, adolescents, infants, or children is not recommended because acute mucocutaneous candidiasis is generally treatable and rarely life-threatening, and because of concerns about the potential development of resistant Candida, possible drug interactions, and the cost of antifungal prophylaxis.
Prevention of Recurrence
Coccidioidomycosis
The USPHS/IDSA recommends that people with HIV who have completed initial treatment for documented coccidioidomycosis should receive long-term suppressive or maintenance therapy (secondary prophylaxis) to help prevent recurrence or relapse. The USPHS/IDSA recommends oral fluconazole as the preferred option, with IV amphotericin B and oral itraconazole (given as capsules) as alternatives. Long-term suppressive therapy to prevent recurrence or relapse of coccidioidomycosis in adults, adolescents, infants, and children with HIV is generally continued for life. Although people with HIV may be at low risk of recurrence of systemic fungal infections if their CD4+ T-cell count rises to greater than 100/mm3 while receiving potent combination antiretroviral therapy, the USPHS/IDSA states that there are not yet enough data to recommend stopping prophylaxis against coccidioidomycosis in these individuals.
Cryptococcosis
The USPHS/IDSA recommends that people with HIV who have completed initial treatment for documented cryptococcosis should receive long-term suppressive or maintenance therapy (secondary prophylaxis) to prevent recurrence or relapse, unless immune recovery has occurred as a result of potent combination antiretroviral therapy. The USPHS/IDSA recommends oral fluconazole as the preferred option, with IV amphotericin B and oral itraconazole (given as capsules) as alternatives. There is some evidence from a randomized, double-blind, controlled study in people with HIV and documented, adequately treated cryptococcal meningitis that oral fluconazole (200 mg once daily) is more effective than oral itraconazole (200 mg once daily) for suppressive therapy in these patients, since the rate of culture-positive relapse of cryptococcosis was 4% in those receiving fluconazole compared with 23% in those receiving itraconazole.

Suppressive or maintenance therapy to prevent recurrence or relapse of cryptococcosis in people with HIV is generally continued for life unless immune recovery has occurred as a result of potent combination antiretroviral therapy. Limited data suggest that stopping suppressive or maintenance therapy in adults and adolescents with HIV who have completed initial treatment for cryptococcosis, remain free of symptoms related to cryptococcosis, and have had sustained increases in CD4+ T-cell counts to greater than 100-200/mm3 in response to potent antiretroviral therapy (for example, for 6 months or longer) is associated with a low risk of recurrence. Based on this and on more extensive cumulative safety data for stopping long-term suppressive therapy for other opportunistic infections, the USPHS/IDSA states that it is reasonable to consider stopping suppressive therapy in individuals who meet these criteria. The USPHS/IDSA notes that recurrences can occur after suppressive therapy is stopped and states that suppressive therapy should be restarted if the CD4+ T-cell count falls to less than 100-200/mm3. The safety of stopping suppressive therapy in infants and children with HIV has not been studied, and children should receive lifelong suppressive therapy after an episode of cryptococcosis.
Histoplasmosis
For long-term suppressive therapy in patients with HIV and documented histoplasmosis that has been adequately treated, the USPHS/IDSA states that oral itraconazole (given as capsules) is the preferred option and IV amphotericin B is an alternative.
Mucocutaneous Candidiasis
The USPHS/IDSA states that long-term suppressive or maintenance therapy should be considered in adults and adolescents with a history of documented esophageal candidiasis, especially those who have had multiple episodes, while taking into account the potential for resistant Candida strains to develop. In addition, the USPHS/IDSA states that suppressive therapy should be considered for infants and children who have severe, recurrent mucocutaneous candidiasis, especially those with esophageal candidiasis. Although many experts do not recommend long-term prophylaxis against recurrent oropharyngeal or vulvovaginal candidiasis in patients with HIV, for the same reasons they do not recommend routine primary prophylaxis against candidiasis, the USPHS/IDSA states that suppressive therapy may be considered for people with HIV who have frequent or severe recurrences of these Candida infections. However, several factors should be considered when deciding on this type of therapy, including how recurrences affect the patient's well-being and quality of life, the need for prophylaxis against other fungal infections, the cost of prophylaxis, drug toxicities, drug interactions, and the potential for drug resistance among Candida and other fungi.
If long-term suppressive therapy to prevent mucocutaneous candidiasis is indicated in adults, adolescents, infants, or children with HIV who have frequent or severe recurrences of oropharyngeal, esophageal, or vaginal candidiasis, the USPHS/IDSA recommends oral fluconazole as the preferred option and itraconazole (given as the oral solution) as an alternative.
Long-term suppressive therapy to prevent recurrence or relapse of fungal infections in patients with HIV generally continues for life. In some patients with HIV who had been receiving oral fluconazole to prevent recurrence of oropharyngeal candidiasis for a median of 18 months (range: 4-98 months) and were receiving potent combination antiretroviral therapy (about 50% had plasma HIV-1 RNA levels below the limit of detection), stopping oral fluconazole suppressive therapy led to recurrence of oropharyngeal candidiasis in only 10% of patients within 6-11 months. Although patients with HIV receiving suppressive antifungal prophylaxis may be at low risk of recurrence of fungal infections if their CD4+ T-cell counts rise to greater than 100/mm3 while receiving potent combination antiretroviral therapy, the USPHS/IDSA states that there are not yet enough data to recommend stopping prophylaxis in these individuals.
Prevention of Fungal Infections in Transplant Patients and Patients with Cancer
Fluconazole is used prophylactically to reduce the incidence of candidiasis in patients undergoing bone marrow transplantation (BMT) who are receiving chemotherapy or radiation therapy. The drug has also been used to prevent fungal infections in patients undergoing liver transplantation and in cancer patients considered at risk for neutropenia and fungal infections. There is some evidence that fluconazole prophylaxis in transplant and cancer patients can reduce the frequency of oropharyngeal and/or systemic candidiasis before neutrophil recovery. In addition, fluconazole prophylaxis may reduce the need for empiric antifungal therapy in these patients. The efficacy of oral fluconazole (400 mg once daily) for prophylaxis against fungal infections in neutropenic patients was evaluated in a randomized, placebo-controlled study involving 274 cancer patients 18-80 years of age receiving cytotoxic chemotherapy or conditioning therapy for BMT. While the percentage of patients not requiring empiric therapy with IV amphotericin B was similar in both groups (57% of those receiving fluconazole and 50% of those receiving placebo required no such therapy), complete success without fungal colonization was achieved in 37% of those receiving fluconazole and 20% of those receiving placebo. In addition, there was a lower incidence of superficial fungal infections in those receiving fluconazole (7%) than in those receiving placebo (18%), and only 3% of those receiving fluconazole developed definite invasive fungal infections compared with 17% of those receiving placebo. While fluconazole prophylaxis did not affect the overall mortality rate, intent-to-treat analysis indicates that the number of deaths attributable to definite invasive fungal infection was lower in the fluconazole group (1 of 15) than in the placebo group (6 of 15).
The use of primary antifungal prophylaxis in cancer patients undergoing myelosuppressive therapy or in patients undergoing BMT or solid organ transplantation remains controversial, particularly because such prophylaxis may predispose the patient to colonization with resistant fungi and/or result in the emergence of highly resistant organisms. Retrospective studies have shown an increased risk of colonization with Candida krusei in BMT recipients and in neutropenic patients who received fluconazole prophylaxis; in one study, about 41% of patients receiving fluconazole had colonization with C. krusei compared with 17% of those not receiving fluconazole. Therefore, most clinicians generally discourage primary prophylactic antifungal therapy except in certain carefully selected high-risk patients in whom the potential benefits are expected to outweigh the possible risks. Many experts, however, state that controlled, randomized studies should continue to evaluate fluconazole's use in preventing fungal infections in cancer patients and in BMT recipients.
Dosage and Administration
Fluconazole is a triazole antifungal used for superficial mucosal candidiasis (oropharyngeal, esophageal, or vaginal) and fungal skin infections. It is also used for systemic infections, including systemic candidiasis, coccidioidomycosis, and cryptococcosis. It has also been tried in blastomycosis, histoplasmosis, and sporotrichosis.
Fluconazole is given by mouth or intravenously in similar doses. For intravenous infusion, it is given as a solution containing 2 mg/mL at a rate of 5 to 10 mL/minute (300 to 600 mL/hour). In Canada, the maximum recommended infusion rate is typically determined by the product monograph in use and institutional practice.
For superficial mucosal candidiasis (other than genital candidiasis), the usual oral dose of Fluconazole in Canada is 50 mg once daily. However, 100 mg once daily may be given if needed.
Treatment usually continues for 7 to 14 days in oropharyngeal candidiasis (except in severely immunocompromised patients), for 14 days in atrophic oral candidiasis associated with dentures, and for 14 to 30 days in other mucosal Candida infections, including esophagitis. Higher doses are recommended in the USA, where an initial dose of Fluconazole 200 mg is followed by 100 mg once daily and where the minimum treatment period is 14 days for oropharyngeal infection, or a minimum of 21 days and at least 14 days after symptoms have resolved for esophageal infections; doses of up to 400 mg once daily may be used for esophageal candidiasis if necessary.
Oral Administration
Fluconazole may be taken by mouth with or without food. Fluconazole powder for oral suspension should be reconstituted when dispensed by adding 24 mL of distilled or purified water to the container holding 0.35 or 1.4 g of the drug to provide a suspension containing 50 or 200 mg/5 mL, respectively. The bottle should be shaken vigorously to suspend the powder, and the suspension should be shaken well just before use.
IV Administration
IV infusions of Fluconazole should be given once daily at a rate not exceeding 200 mg/hour. Fluconazole injections for IV infusion should be visually inspected for discolouration and particulate matter before administration whenever the solution and container permit. The injection for IV infusion should be discarded if the solution is cloudy or has precipitated, or if the seal is not intact. Viaflex® Plus containers of Fluconazole should be checked for minute leaks by firmly squeezing the bag. The injection should be discarded if the container seal is not intact, leaks are found, or the solution is cloudy or contains a precipitate. Additives should not be introduced into the plastic injection container. The injection in plastic containers should not be used in series connections with other plastic containers, since this could result in air embolism from residual air being drawn from the primary container before administration from the secondary container is complete.
Fluconazole 150 mg as a single oral dose may be used for genital candidiasis (vaginal candidiasis or candidal balanitis). Dermatophytosis, pityriasis versicolor, and Candida skin infections may be treated with Fluconazole 50 mg taken by mouth once daily for up to six weeks.
Systemic candidiasis, cryptococcal meningitis, and other cryptococcal infections may be treated by mouth or by intravenous infusion with Fluconazole. The initial dose is 400 mg, followed by 200 to 400 mg once daily. The duration of therapy is based on clinical and mycological response, but it is usually at least 6 to 8 weeks. In Canada, treatment of cryptococcal meningitis is generally continued for 10 to 12 weeks after the CSF cultures become negative.
Fluconazole may also be used in daily doses of 100 to 200 mg by mouth or intravenously to prevent relapse after a primary course of antifungal treatment for acute cryptococcal meningitis in patients with AIDS. In immunocompromised patients at risk of fungal infections, Fluconazole may be given prophylactically by mouth or by intravenous infusion at 50 to 400 mg once daily. However, long-term prophylaxis has been associated with the emergence of resistant organisms. Doses for children over 4 weeks of age are 3 mg/kg/day for superficial infections (a loading dose of 6 mg/kg may be used on the first day if necessary) and 6 to 12 mg/kg/day for systemic infections. A dose of 3 to 12 mg/kg/day may be given for prophylaxis in immunocompromised children. For infants under 2 weeks of age, all of these doses should be given once every 72 hours. For those aged between 2 and 4 weeks, the doses should be given every 48 hours.
A maximum dose of 400 mg once daily should not be exceeded in children, or 12 mg/kg at appropriate intervals in infants. Dosage may need to be reduced in patients with renal impairment.
High Doses
Higher-than-labelled doses of Fluconazole have been tried in patients with life-threatening infections caused by Candida spp., Cryptococcus neoformans, and Coccidioides immitis.
Dose-finding studies have found daily doses of 800 to 1000 mg of Fluconazole to be effective and well tolerated. In a study of 11 patients with HIV who received fluconazole 800 to 1000 mg once daily intravenously for 3 weeks, then by mouth until the CSF culture became negative, 6 patients had responded at 10 weeks, and another 2 improved clinically. Daily doses of up to 800 mg have been used in blastomycosis and coccidioidomycosis, and doses of 10 mg/kg/day have been tried in disseminated candidiasis.
Intermittent Doses
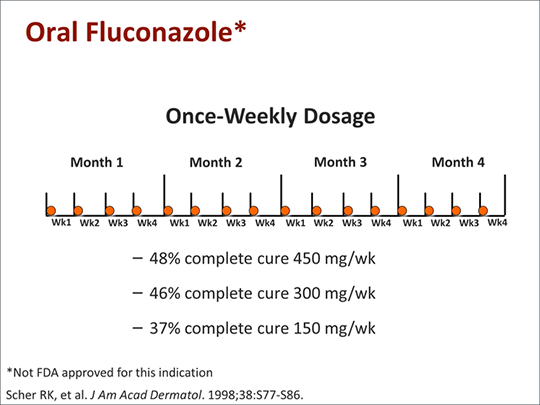
Concern has been raised about the increasingly widespread use of Fluconazole, particularly the effect of continuous fluconazole therapy in immunocompromised patients on the development of resistance. Nevertheless, Fluconazole remains popular for primary and secondary prophylaxis. Some investigators have suggested intermittent dosing, although this could further increase the risk of infections with resistant organisms. Once-weekly treatment with Fluconazole has been tried in onychomycosis and tinea capitis.
Administration in Renal Impairment
Patients with renal impairment may require a lower dose. Normal loading or initial doses of Fluconazole should be given on the first day of treatment, and subsequent doses should be adjusted according to creatinine clearance (CC):
- CC more than 50 mL/minute: 100% of the standard recommended dose;
- CC less than 50 mL/minute and not receiving dialysis: 50% of the standard recommended dose;
- patients on regular hemodialysis: 100% of the standard recommended dose after each dialysis session. No dosage adjustment is needed in patients with renal impairment given single-dose therapy.
Adult Dosage
Oropharyngeal and Esophageal Candidiasis
For treating oropharyngeal or esophageal candidiasis, the usual adult dosage of Fluconazole is 200 mg as a single dose on the first day of therapy, followed by 100 mg or 200 mg once daily. Doses up to 400 mg once daily may be used depending on the patient's response. Although clinical signs of oropharyngeal candidiasis generally resolve within several days after starting fluconazole therapy, the manufacturer and some clinicians recommend continuing the drug for at least 2 weeks to reduce the chance of relapse.

However, other clinicians question the need for prolonged therapy in patients with this infection. Patients with esophageal candidiasis should receive fluconazole therapy for at least three weeks and for at least two weeks after symptoms have resolved. The optimal dosage for maintenance therapy in patients with oropharyngeal candidiasis has not been established. Oral doses of 50-100 mg once daily have generally been used effectively for maintenance therapy in these patients; doses up to 200 mg once daily have occasionally been used.
Vulvovaginal Candidiasis
For the treatment of uncomplicated vulvovaginal candidiasis in nonpregnant women, the usual dosage of oral Fluconazole is a single 150-mg oral dose. For the treatment of recurrent vulvovaginal candidiasis in nonpregnant women, two 150-mg doses of oral Fluconazole should be given 3 days apart to achieve mycologic remission. Then, a maintenance regimen of 100-150 mg once weekly should be given for 6 months to prevent recurrence. A 2-dose regimen of oral Fluconazole (two 150-mg doses given 3 days apart) is also recommended for the treatment of severe vulvovaginal candidiasis in nonpregnant women.
Leishmaniasis
Fluconazole has been tried for the treatment of cutaneous leishmaniasis caused by Leishmania major. In a randomized, double-blind, placebo-controlled study, 80 patients received a six-week course of oral Fluconazole 200 mg once daily, of whom 63 had complete healing of lesions after 3 months, compared with 22 of 65 patients who received placebo. However, others have reported a response rate that was not significantly different from placebo.
Other Candidal Infections
For treating systemic candidiasis, the usual adult dosage of Fluconazole is 400 mg as a single dose on the first day of therapy, followed by 200 mg once daily. In a limited number of patients with candidal urinary tract infections and peritonitis, doses of 50-200 mg once daily have been used. The optimum dosage and duration of therapy in patients with candidemia, disseminated candidiasis, and pneumonia have not been established; however, a limited number of such patients have received fluconazole doses of up to 400 mg once daily. Some clinicians have recommended that patients with invasive candidiasis receive Fluconazole in a dosage of 400-800 mg once daily. Therapy should be continued for at least 4 weeks and for at least 2 weeks after symptoms have resolved.
Cryptococcal Infections
To treat cryptococcal meningitis, the usual adult dosage of Fluconazole is 400 mg as a single dose on the first day of therapy, followed by 200 to 400 mg once daily. Some evidence suggests that the 400-mg dosage is more effective than lower dosages in treating this infection. A higher dosage of Fluconazole (i.e., 800-1000 mg once daily) has been used in some patients with human immunodeficiency virus (HIV) infection for the treatment of cryptococcal meningitis. For initial therapy of cryptococcal meningitis, Fluconazole usually is continued for 10-12 weeks after the CSF is sterile.

Coccidioidomycosis
For treating coccidioidal meningitis in adults, fluconazole doses of 200-800 mg once daily have been recommended. For the treatment of coccidioidal meningitis in patients with AIDS, fluconazole doses of 400-800 mg once daily are recommended. Concomitant intracisternal, intraventricular, or intrathecal amphotericin B therapy has been used in some patients.
Blastomycosis or Histoplasmosis
If Fluconazole is used to treat blastomycosis or histoplasmosis, a daily dosage of 400-800 mg is recommended.
Prevention of Fungal Infections in HIV-infected Individuals
In Canadian HIV care, primary prophylaxis against cryptococcosis in adults or adolescents with HIV infection and absolute helper/inducer (CD4+, T4+) T-cell counts less than 50/mm3 is not routinely recommended, but selected high-risk patients may be considered for oral Fluconazole 100-200 mg once daily based on individual clinical circumstances.
While there is some evidence that oral Fluconazole given at 400 mg once weekly may be effective for primary prophylaxis against fungal infections in people with HIV, this regimen is not included in current USPHS/IDSA guidelines. Further study is needed to evaluate the efficacy of regimens other than daily administration for primary prophylaxis.
For long-term suppressive or maintenance therapy (secondary prophylaxis) of coccidioidomycosis in adults or adolescents with HIV who have had documented, adequately treated infections, the USPHS/IDSA recommends fluconazole 400 mg once daily. For suppressive or maintenance therapy to prevent recurrence or relapse of cryptococcosis in adults or adolescents with HIV who have had documented, adequately treated infections, the usual dosage of oral Fluconazole is 200 mg once daily.
Some clinicians recommend that, for long-term suppressive therapy against cryptococcosis, oral Fluconazole be given at 400 mg once daily for the first 4 weeks, followed by 200 mg once daily. If oral Fluconazole is used for long-term suppressive or maintenance therapy (secondary prophylaxis) to prevent recurrence or relapse of mucocutaneous candidiasis (oropharyngeal, vaginal, esophageal) in adults or adolescents with HIV who have had frequent or severe episodes of these Candida infections, the USPHS/IDSA recommends a dosage of 100-200 mg once daily.
While Fluconazole has been used at a dose of 200 mg once weekly for long-term suppressive therapy in women with HIV who have a history of oropharyngeal or vaginal candidiasis, this regimen is not included in current USPHS/IDSA guidelines. There are concerns that this approach could promote the emergence of fluconazole-resistant Candida strains. Long-term suppressive or maintenance therapy to prevent recurrence or relapse of fungal infections in patients with HIV is generally continued for life. However, the USPHS/IDSA states that it may be reasonable to stop suppressive or maintenance therapy for cryptococcosis in certain adults and adolescents whose immune function has recovered with potent combination antiretroviral therapy.
Prevention of Fungal Infections in Transplant Patients and Patients with Cancer
To prevent candidiasis in bone marrow transplant recipients, the recommended dose of Fluconazole is 400 mg once daily. In patients expected to develop severe granulocytopenia (neutrophil count less than 500/mm3), fluconazole should be started several days before the anticipated onset of neutropenia. Treatment should continue for 7 days after the neutrophil count rises above 1000/mm3.
Pediatric Dosage
The usual dose of Fluconazole in pediatric patients ranges from 3-12 mg/kg once daily; doses above 600 mg daily are not recommended. The manufacturer states that doses of 3, 6, or 12 mg/kg daily in pediatric patients are equivalent to 100, 200, or 400 mg daily, respectively, in adults. Some older children may clear the drug at rates similar to adults.
To treat meningitis or septicemia caused by susceptible Candida, neonates and infants 3 months of age or younger have received Fluconazole at a dose of 5-6 mg/kg once daily, given by mouth or by IV infusion over 1 hour. In some neonates and infants with septicemia, an initial loading dose of 10 mg/kg was given, followed by 5 mg/kg once daily. Based on available pharmacokinetic data for Fluconazole in premature neonates, the manufacturer recommends that neonates 2 weeks of age or younger receive the same daily dose as older children. However, the dose should be given once every 72 hours.
Oropharyngeal and Esophageal Candidiasis
For the treatment of oropharyngeal or esophageal candidiasis, the manufacturer recommends that pediatric patients receive 6 mg/kg of Fluconazole on the first day, followed by 3 mg/kg once daily. The dose for esophageal candidiasis may be increased up to 12 mg/kg daily if needed, based on the patient's condition and response to the drug. Treatment for oropharyngeal candidiasis should continue for at least 2 weeks to reduce the chance of relapse. Treatment for esophageal candidiasis should continue for at least 3 weeks and for at least 2 weeks after symptoms have resolved.
Other Fungal Infections
Fluconazole has been given at doses of 6-12 mg/kg daily to treat systemic candidal infections in pediatric patients. The manufacturer recommends treating cryptococcal meningitis in pediatric patients with an initial 12 mg/kg dose on the first day, followed by 6 mg/kg once daily. If needed, the dose may be increased to 12 mg/kg daily based on the patient's condition and response to the drug. Fluconazole should be continued for 10-12 weeks after the CSF becomes culture-negative.
Prevention of Fungal Infections in HIV-infected Individuals
The recommended oral dose of Fluconazole for primary prophylaxis against cryptococcosis in infants and children with HIV and severe immunosuppression is 3-6 mg/kg once daily. If oral Fluconazole is used for long-term suppressive or maintenance therapy to prevent recurrence or relapse of cryptococcosis or mucocutaneous candidiasis (oropharyngeal, esophageal) in infants and children with HIV, the recommended dose is 3-6 mg/kg once daily. A dose of 6 mg/kg once daily is recommended to prevent recurrence or relapse of coccidioidomycosis in these pediatric patients.
Interactions
In general, Fluconazole has fewer interactions than Itraconazole or ketoconazole. Using rifampicin with Fluconazole lowers plasma concentrations of Fluconazole. Hydrochlorothiazide with Fluconazole has led to clinically insignificant increases in plasma fluconazole concentrations.
Fluconazole may interfere with the metabolism of some other drugs, mainly by inhibiting the cytochrome P450 isoenzymes CYP3A4 and CYP2C9. This may explain reported increases in plasma concentrations of bosentan, ciclosporin, midazolam, nevirapine, amitriptyline, nortriptyline, phenytoin, rifabutin, sulfonylurea hypoglycemics and nateglinide, selective cyclooxygenase-2 inhibitors such as celecoxib and parecoxib, tacrolimus, triazolam, warfarin, and zidovudine. Fluconazole may inhibit the formation of a toxic metabolite of sulfamethoxazole. Higher terfenadine concentrations after high doses of Fluconazole have been associated with ECG abnormalities.
A similar effect may be expected with astemizole. However, using Fluconazole with cisapride could increase cisapride concentrations and cause related toxicity, so this combination should be avoided. Fluconazole should also be avoided with astemizole, cisapride, or terfenadine because of the risk of cardiac arrhythmias. Syncope has been attributed to increased amitriptyline concentrations when amitriptyline was given with Fluconazole.
Fluconazole may also reduce the clearance of theophylline. Concentrations of contraceptive steroids have been reported to both increase and decrease in patients taking Fluconazole, and the effectiveness of oral contraceptives may be affected. For more information on interactions between azoles and drugs metabolized by the cytochrome P450 isoenzyme CYP3A, see under Itraconazole.
Fluoroquinolones
Both levofloxacin and fluconazole can prolong the QT interval. The simultaneous use of intravenous levofloxacin and Fluconazole led to an episode of torsade de pointes in a patient on hemodialysis.

Microbiological Interactions
An in vitro synergistic antifungal effect was seen with terbinafine and Fluconazole against strains of Candida albicans.
Resistance
The emergence of Fluconazole-resistant Candida spp. strains has become increasingly important, especially in immunocompromised patients receiving long-term prophylaxis with Fluconazole. In addition to resistance in Candida albicans, infections with Candida dubliniensis, Candida glabrata, and Candida krusei have also been noted in these patients, and all may be less sensitive to Fluconazole than Candida albicans. Secondary resistance of Candida glabrata has been reported during fluconazole therapy.
Resistance to Fluconazole has been reported more often than resistance to ketoconazole or Itraconazole, which may be related to its widespread use. Cross-resistance with other azoles and with amphotericin B has also been reported.
Fluconazole resistance has also been reported in Cryptococcus neoformans and Histoplasma capsulatum. Histoplasmosis developed during treatment with Fluconazole in a patient with HIV infection. Fluconazole-resistant C. neoformans was isolated from an immunocompetent patient who had not previously been exposed to azole antifungals.
Fluconazole Drug-Drug Interactions
Alfentanil
In a randomized, double-blind, placebo-controlled, crossover study in nine subjects, Fluconazole 400 mg reduced the clearance of alfentanil 20 micrograms/kg by 55% and increased alfentanil-induced subjective effects.
Amitriptyline
An interaction between Fluconazole and amitriptyline has been reported.
- A 12-year-old boy with prostatic rhabdomyosarcoma had periodic episodes of syncope over 7 months while taking Fluconazole for chemotherapy-induced mucositis. He had taken Fluconazole before without problems. However, he had also been taking a stable dose of amitriptyline for neuropathic pain. After amitriptyline was stopped, he had no further episodes. The effect was confirmed on re-challenge.
Concurrent use of Fluconazole probably increases exposure to amitriptyline. Three case reports in adults have shown increased amitriptyline plasma concentrations with concurrent fluconazole use; in one patient, a 57-year-old woman, the QT interval was prolonged and torsade de pointes occurred.
Amphotericin
In vitro studies and animal experiments have produced conflicting results about possible antagonism between the effects of Fluconazole and amphotericin on Candida species. However, large randomized, double-blind comparisons of Fluconazole with and without amphotericin for 5 days in non-neutropenic patients with candidemia showed no evidence of antagonism, but did show faster clearance of the organism from the blood and a trend toward improved outcomes in those who received the combination.
Antacids
Fluconazole absorption after oral administration is not affected by gastric pH, so antacids such as co-magaldrox do not affect it.
Antihistamines
Using terfenadine with Fluconazole can lead to dangerously high terfenadine concentrations, resulting in cardiotoxicity. The same effect is suspected with astemizole.
Benzodiazepines
Twelve healthy men took part in a randomized, double-blind, four-way crossover study to assess the interaction between Fluconazole and bromazepam. They received single oral or rectal doses of bromazepam (3 mg) after a 4-day pretreatment with oral Fluconazole (100 mg/day) or placebo. Fluconazole caused no significant changes in the pharmacokinetics or pharmacodynamics of oral or rectal bromazepam.
Fluconazole increased blood concentrations of midazolam and triazolam.
The effects of Fluconazole (400 mg loading dose followed by 200 mg/day) on the kinetics of midazolam were studied in 10 mechanically ventilated adults receiving a stable infusion of midazolam. Midazolam concentrations increased up to four-fold after fluconazole therapy started; these changes were most pronounced in patients with renal insufficiency. During the study, the ratio of a-hydroxy midazolam to midazolam progressively fell. The authors concluded that in ICU patients receiving Fluconazole, a reduction in the midazolam dose should be considered if sedation increases.
In a study of the pharmacokinetics and pharmacodynamics of oral midazolam 7.5-15 mg, switching from inhibition of metabolism by Itraconazole 200 mg/day to induction of metabolism by rifampicin 600 mg/day caused up to a 400-fold change in the AUC of oral midazolam.
Calcium Channel Blockers
Fluconazole enhanced the blood pressure-lowering effects of nifedipine by increasing its plasma concentrations in a 16-year-old patient with malignant pheochromocytoma who was taking chronic nifedipine for hypertension and was given Fluconazole for Candida septicemia.
Carbamazepine
Fluconazole can cause carbamazepine toxicity, presumably by inhibiting CYP3A4.
- A 33-year-old man on stable therapy with carbamazepine (400 mg three times daily) for a seizure disorder became stuporous due to carbamazepine toxicity after taking Fluconazole 150 mg/day for 3 days. Withdrawal of both drugs resulted in a fall in carbamazepine concentrations (maximum concentration 25 µg/ml) and a return to the patient's baseline mental status. Carbamazepine was restarted, and the patient had no further adverse events.
- Carbamazepine serum concentrations increased during concomitant fluconazole administration (400 mg/day) in a 38-year-old man.
Ciclosporin
Fluconazole can increase ciclosporin concentrations by inhibiting CYP3A4. In some studies, minimal or no effects were seen, while in others Fluconazole increased ciclosporin concentrations. Differences in the dose and duration of fluconazole treatment may explain these discrepancies. For example, there was no interaction at a fluconazole dose of 100 mg/day, but higher doses of Fluconazole (400 mg/day or more) increased blood ciclosporin and tacrolimus concentrations.
The interaction between ciclosporin and Fluconazole was evaluated retrospectively in 19 kidney and pancreas/kidney transplant recipients. Both intravenous and oral Fluconazole changed blood ciclosporin concentrations. Five subjects did not have a significant interaction, and 15 did. No patient had nephrotoxicity or transplant rejection related to antifungal therapy.
The effects of higher doses of Fluconazole on ciclosporin immunosuppression were investigated in six renal transplant patients in a prospective, unblinded, crossover study. Baseline renal function, ciclosporin AUC, Cmax, Cmm, fmax, and clearance were compared with values measured 2,4 and 7 days after starting oral Fluconazole at a dose of 200 mg/day. From day 8 onward, the ciclosporin dose was reduced by 50%, and the above parameters were measured again on day 14. The results are shown in Table 1. On repeated-measures ANOVA, only the AUC and Cmax on day 4 of Fluconazole were significantly higher than on day 0. There were no significant changes in ciclosporin clearance or fmax. The authors concluded that changes in Cmin may not be sensitive enough to detect the interaction described and suggested monitoring the AUC near day 4 of treatment to guide ciclosporin dose adjustments in all patients taking concomitant Fluconazole.
Cimetidine
Fluconazole absorption after oral administration is not affected by gastric pH, so cimetidine has no effect.
Clarithromycin
The effects of fluconazole and clarithromycin on the pharmacokinetics of rifabutin and 25-O-desacetylrifabutin were studied in 10 patients with HIV who received rifabutin 300 mg daily in addition to fluconazole 200 mg daily and clarithromycin 500 mg daily. There was a 76% increase in the plasma AUC of rifabutin when either fluconazole or clarithromycin was given alone and a 152% increase when both drugs were given together. The authors concluded that patients should be monitored for adverse effects of rifabutin when it is given together with fluconazole or clarithromycin.
Cyclophosphamide
Cyclophosphamide is a prodrug that is metabolized by CYP450 enzymes to form cytotoxic alkylating species, and the extent of cyclophosphamide metabolism correlates with both treatment effectiveness and toxicity. In vitro studies using six human liver microsome samples showed that the IC50 of fluconazole for reducing the production of 4-hydroxycyclophosphamide was 9-80 micromol/L.
A retrospective study in 22 children with cancer examined the potential interaction between fluconazole and cyclophosphamide. Children with an established cyclophosphamide metabolism profile who were not receiving other drugs known to affect drug metabolism were selected; 9 were receiving fluconazole and 13 served as controls. Plasma clearance was significantly lower in patients receiving concomitant fluconazole (2.4 versus 4.2 L/hour/m²). It is unclear whether this interaction reduces the therapeutic effectiveness of cyclophosphamide.
Doxorubicin
The effect of Fluconazole on the plasma pharmacokinetics of doxorubicin was investigated in a randomized crossover study in non-human primates. Fluconazole (10 mg/kg/day) was given intravenously for 4 days before doxorubicin (2.0 mg/kg intravenously). Pretreatment with Fluconazole did not affect the pharmacokinetics of doxorubicin, and the incidence of severe neutropenia (absolute neutrophil count below 0.5 x 109/1) was higher with doxorubicin alone than with the combination of doxorubicin and Fluconazole. Therefore, Fluconazole does not appear to contribute to the marrow-suppressive effects of doxorubicin.
Flucytosine
Using Fluconazole and flucytosine together may have an additive effect. This combination may help in the treatment of cryptococcal meningitis.
HIV Protease Inhibitors
The pharmacokinetic interaction of fluconazole 400 mg once daily and indinavir 1000 mg three times daily was evaluated in an 8-day, placebo-controlled crossover study; no significant interaction was observed.
The effect of Fluconazole on the steady-state pharmacokinetics of ritonavir and saquinavir was studied in patients with HIV-1. They received the protease inhibitor (saquinavir 1200 mg three times daily, n = 5, or ritonavir 600 mg twice daily) alone on day 1 and then with Fluconazole 400 mg on day 2 and 200 mg on days 3-8. The median increase in saquinavir AUC was 50%, and the median increase in Cmax was 56%. In contrast, Fluconazole did not affect the disposition of ritonavir.
Methadone
An interaction between Fluconazole and methadone (a substrate of CYP3A4, CYP2C9, and CYP2C) has been reported.
While taking a stable dose of methadone, a 60-year-old man with advanced cancer developed respiratory depression 2 days after receiving intravenous Fluconazole for refractory oral candidiasis. Intravenous naloxone reversed the respiratory depression.
In a randomized, double-blind, placebo-controlled study in 25 patients, Fluconazole 200 mg/day increased methadone concentrations, but patients treated with Fluconazole did not have signs or symptoms of significant narcotic overdose.
Omeprazole
Fluconazole absorption after oral administration is not affected by gastric pH, so omeprazole has no effect. Omeprazole is extensively metabolized in the liver by 5-hydroxylation and sulfoxidation reactions, catalyzed mainly by CYP2C19 and CYP3A4, respectively. Fluconazole is a potent competitive inhibitor of CYP2C19 and a weak inhibitor of CYP3A4. The effect of Fluconazole on the pharmacokinetics of a single oral dose of omeprazole 20 mg was evaluated after a single oral dose of Fluconazole 100 mg and after 4 days of oral administration of 100 mg/day in 18 healthy male volunteers. Fluconazole increased the Cmax and mean AUC of omeprazole and prolonged its half-life (2.59 versus 0.85 hours).
Oral Contraceptives
Fluconazole did not significantly alter the pharmacokinetics of ethinylestradiol or norgestrel, and the investigators interpreted this as suggesting that treatment with Fluconazole in users of oral contraceptives would not increase the risk of pregnancy. However, the study used a 50 mg dose of Fluconazole, and experience with higher doses has shown different results.
In another study, Fluconazole reduced the systemic availability of ethinylestradiol in an oral contraceptive, but the doses used were not reported.
In an open, crossover study of 10 young, healthy subjects, Fluconazole 150 mg increased serum concentrations of ethinylestradiol 30-35 µg/day.
The potential pharmacokinetic interaction between Fluconazole 300 mg once weekly and an oral contraceptive containing ethinylestradiol and norethindrone (Ortho-Novum 7/7/7; Ortho-McNeil Pharmaceutical Inc, Raritan, NJ) was studied in a placebo-controlled, double-blind, randomized, two-way crossover study in 26 healthy women aged 18-36 years. During the first cycle, they took the oral contraceptive only. During the second cycle, they were randomly assigned to oral contraceptive + Fluconazole or oral contraceptive + placebo. In the third cycle, they received the alternate treatment.
Fluconazole caused small but statistically significant increases in the AUCo-24 of ethinylestradiol and norethindrone. Those given Fluconazole experienced no treatment-related side effects.
It therefore appears that there is no threat of contraceptive failure due to concomitant fluconazole administration.
Phenytoin
Co-administration of Fluconazole and phenytoin resulted in markedly higher phenytoin concentrations.
Rifamycins
The combination of rifampicin with Fluconazole has insignificant effects.
Statins
The effects of fluconazole on plasma fluvastatin and pravastatin concentrations were studied in two separate randomized, double-blind, two-phase crossover studies. Healthy volunteers received oral fluconazole (400 mg on day 1 and 200 mg on the following days) or placebo. On day 4, they received a single oral dose of fluvastatin 40 mg or pravastatin 40 mg.
Fluconazole increased the plasma AUC and half-life of fluvastatin by 80% but had no significant effects on the pharmacokinetics of Pravastatin. The mechanism behind the prolonged elimination of fluvastatin was probably inhibition of CYP2C9. In contrast, Pravastatin does not appear to be susceptible to interactions with Fluconazole and other CYP2C9 inhibitors.
The effect of Fluconazole on the pharmacokinetics of rosuvastatin was investigated in a randomized, double-blind, two-way, crossover, placebo-controlled study. Healthy male volunteers were given 200 mg/day fluconazole or matching placebo for 11 days; rosuvastatin 80 mg was co-administered on day 8. Plasma concentrations of rosuvastatin, N-desmethyl rosuvastatin, and active and total HMG-CoA reductase inhibitors were measured up to 96 hours after the dose.
Fluconazole increased the AUC and Cmax of rosuvastatin by 14% and 9%, respectively. Limited data for the N-desmethylated metabolite showed that the Cmax fell by about 25%. Fluconazole did not affect the proportion of circulating active or total HMG-CoA reductase inhibitors accounted for by circulating rosuvastatin. Therefore, Fluconazole produced only minor changes in rosuvastatin kinetics, which were not clinically relevant.
Sulfonylureas
Concurrent administration of Fluconazole with tolbutamide resulted in increased tolbutamide concentrations.
- A 56-year-old HIV-positive patient with diabetes mellitus taking gliclazide 160 mg/day developed severe hypoglycemia when treated with co-trimoxazole 480 mg/day and Fluconazole 200 mg/day. The authors speculated that Fluconazole may have inhibited gliclazide metabolism by inhibiting CYP2C9.
The effects of fluconazole and fluvoxamine on the pharmacokinetics and pharmacodynamics of glimepiride were studied in a randomized, double-blind, crossover study in 12 healthy volunteers who received fluconazole 200 mg/day, fluvoxamine 100 mg/day, or placebo once daily for 4 days. On day 4, they received a single oral dose of glimepiride 0.5 mg.
Fluconazole increased the mean total AUC of glimepiride to 238%, the peak plasma concentration to 151% of control values, and prolonged the half-life of glimepiride from 2.0 to 3.3 hours. This was probably due to inhibition of CYP2C9-mediated biotransformation of glimepiride by Fluconazole. However, Fluconazole did not cause statistically significant changes in the effects of glimepiride on blood glucose concentrations.
Tacrolimus
Since tacrolimus is metabolized by intestinal and hepatic CYP3A4, drugs that inhibit CYP3A4 can reduce tacrolimus metabolism and increase tacrolimus blood concentrations.
The effect of Fluconazole on tacrolimus blood concentrations was investigated in eight liver transplant patients in whom prophylactic Fluconazole (200 mg/day) was withdrawn because of rises in hepatic transaminases, renal dysfunction, or eosinophilia (n = 1 each). Calculated tacrolimus concentrations fell by 13-81% (median 41%) between the fourth and ninth days after Fluconazole was withdrawn. Tacrolimus blood concentrations should be monitored carefully, and doses should be increased as needed after Fluconazole is stopped.
The interaction between tacrolimus and Fluconazole was evaluated retrospectively in 19 kidney and pancreas/kidney transplant recipients. Both intravenous and oral Fluconazole changed tacrolimus blood concentrations. Five subjects did not have a significant interaction, and 15 did. No patient had nephrotoxicity or transplant rejection related to antifungal therapy.
- A 17-year-old man with cystic fibrosis who took Itraconazole after a lung-liver transplant had high trough concentrations of tacrolimus despite the relatively low dose (0.1-0.3 mg/kg/day).
- A patient taking tacrolimus 0.085 mg/kg twice daily with itraconazole 200-400 mg/day developed ketoacidosis, neutropenia, and thrombocytopenia, requiring withdrawal of both drugs.
- A 34-year-old renal transplant recipient taking a stable regimen of tacrolimus and methylprednisolone was given itraconazole 100 mg twice daily for a urinary tract yeast infection. Concomitant therapy with Itraconazole led to a marked increase in tacrolimus trough concentrations on the second day of therapy (from 13 to 21 µg/ml) and an increase in serum creatinine concentrations, requiring a 50% reduction in the tacrolimus dose.
When Itraconazole was withdrawn, its effect on tacrolimus kinetics took 12 days to reverse. The inhibitory effect of Itraconazole occurred quickly, while it took much longer to wear off, which is important for clinical management. Therefore, during co-administration of Itraconazole with tacrolimus, close monitoring of tacrolimus blood concentrations and careful dose adjustments are essential to avoid toxicity.
Warfarin
Co-administration of Fluconazole and warfarin has, in some cases, led to prolongation of the prothrombin time.
Zidovudine
The combination of Fluconazole with valproic acid inhibited zidovudine glucuronidation in human hepatic microsomes in vitro more than other drugs, such as atovaquone and methadone.
Adverse Effects
Read the indications for use if you want to order Fluconazole online.
Side effects reported with Fluconazole most often affect the gastrointestinal tract and include abdominal pain, diarrhea, flatulence, nausea and vomiting, and taste disturbance. Other reported side effects include headache, dizziness, leukopenia, thrombocytopenia, hyperlipidemias, and elevated liver enzyme levels. Severe hepatotoxicity has been reported in patients with serious underlying illnesses such as AIDS or malignancy. Anaphylaxis and angioedema have been reported rarely. Skin reactions are uncommon, but exfoliative skin reactions such as toxic epidermal necrolysis and Stevens-Johnson syndrome have occurred, more often in patients with AIDS.
Alopecia
Alopecia has occasionally been reported in patients receiving Fluconazole, especially during prolonged use.
Effect on Electrolyte Balance
Hypokalemia was associated with Fluconazole in 3 patients with acute myeloid leukemia.
Effects on the Heart
QT prolongation and torsade de pointes have been reported rarely in patients receiving Fluconazole.
Effects on the Liver
Although severe hepatic reactions to Fluconazole are rare, they have been reported, especially in patients with serious underlying illness or hepatic dysfunction. Elevated liver enzymes are common, and jaundice has also been reported. Hepatic necrosis has rarely been seen post-mortem in patients with severe underlying disease who had received Fluconazole. In one such patient, hepatotoxicity was considered dose-dependent.

Hypersensitivity
Desensitization has been carried out successfully in a patient with AIDS who showed hypersensitivity to both Fluconazole and Itraconazole. Over 7 days, gradually increasing oral doses of Fluconazole were given, starting at 5 mg daily; after that, the dose was maintained at 400 mg daily. No side effects were noted during the desensitization period or in the 3 months before the report was published.
Precautions
Fluconazole should be used with caution in patients with impaired liver or kidney function. Abnormalities in hematologic, hepatic, and renal function tests have been seen in patients with serious underlying illnesses such as AIDS or malignancy. Cases of torsade de pointes and QT prolongation have been reported rarely, and caution is advised when giving Fluconazole to patients with proarrhythmic conditions. Teratogenicity has occurred in animals given high doses of Fluconazole, and its use is not recommended during pregnancy.
Breastfeeding
Fluconazole passes into breast milk, reaching concentrations similar to those found in maternal plasma. However, licensed product information does not recommend its use in breastfeeding women. In one report, no harmful effects other than a slight increase in lactate dehydrogenase level were seen in an infant exposed to Fluconazole in breast milk for 6 weeks. In Canadian clinical practice, fluconazole is generally considered compatible with breastfeeding when clinically indicated.
Pregnancy
High (toxic) doses of Fluconazole, Itraconazole, and ketoconazole have been reported to be teratogenic in rodents.
Although there is limited information about the use of these drugs in human pregnancy, there is a report of a woman who took Fluconazole 400 mg daily throughout pregnancy and gave birth to an infant with severe craniofacial and limb abnormalities. The abnormalities resembled those associated with Antley-Bixler syndrome, a genetic disorder. However, a teratogenic effect could not be excluded.
Although prescription-event-monitoring studies of Fluconazole did not identify adverse effects on the fetus, congenital abnormalities have occurred in infants whose mothers were given high doses of Fluconazole for 3 months or more. Data collected by the manufacturer on 198 women exposed to Itraconazole during the first trimester of pregnancy indicated that the malformation rate in both exposed women and matched controls was within the expected baseline risk for the general population.
Nevertheless, manufacturers recommend avoiding Fluconazole, Itraconazole, and ketoconazole during pregnancy. Licensed product information states that doses of voriconazole equivalent to those used therapeutically are teratogenic and embryotoxic in rodents. Therefore, the manufacturer recommends avoiding voriconazole during pregnancy and that women of child-bearing potential use effective contraception during treatment.
Similar recommendations have been made for posaconazole. Other azole antifungals, including butoconazole, clotrimazole, econazole, miconazole, sulconazole, terconazole, and tioconazole, are reported to be embryotoxic but not teratogenic in rodents given high doses. Many of these drugs are used topically or intravaginally, and systemic absorption from these routes varies. While these drugs may not necessarily be contraindicated in pregnancy, these potential risks should be considered when choosing antifungal therapy for such patients.














